From Wikipedia, the free encyclopedia
Content deleted Content added
|
|
|||
| Line 7: | Line 7: | ||
|
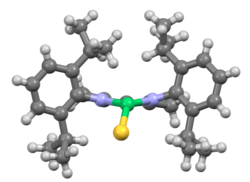
[[File:FUYTEAfragment.png|Diisopropyl substituents make up most of this [[NacNac]] ligand.|thumb]] |
[[File:FUYTEAfragment.png|Diisopropyl substituents make up most of this [[NacNac]] ligand.|thumb]] |
||
|
The diisopropylphenyl structure |
The diisopropylphenyl structure of [[alkane]]-substituted [[benzene]], so it is not very chemically reactive. It serves instead to provide [[steric hindrance]], limiting reactions of other nearby functional groups. |
||
|
The diisopropylphenyl structure is found in many ligands calling for steric bulk. [[NacNac]] ligands can be prepared from [[2,6-Diisopropylaniline|2,6-diisopropylaniline]]. [[Terphenyl]] ligands featuring two diisopropylphenyl substituents, systematically bis(diisopropylphenyl)phenyl, are capable of stabilising high-[[bond order]] compounds such as [[dimetallene]]s.<ref>{{cite journal|last1=Rivard |first1=Eric |last2=Power |first2=Philip P. |title=Multiple Bonding in Heavier Element Compounds Stabilized by Bulky Terphenyl Ligands |journal=Inorganic Chemistry |date=2007 |volume=46 |issue=24 |pages=10047–10064 |doi=10.1021/ic700813h |pmid=17975890 }}</ref> The first stable [[digermyne]] was prepared using DIPP-based terphenyl ligands.<ref>{{cite journal|last1= Stender|first1= Matthias|last2= Phillips|first2= Andrew D.|last3= Wright|first3= Robert J.|last4= Power|first4= Philip P.|title= Synthesis and Characterization of a Digermanium Analogue of an Alkyne|journal= Angewandte Chemie International Edition|date= 2002|volume= 41|issue= 10|pages= 1785–1787|doi= 10.1002/1521-3773(20020517)41:10<1785::AID-ANIE1785>3.0.CO;2-6|pmid= 19750717}}</ref> |
The diisopropylphenyl structure is found in many ligands calling for steric bulk. [[NacNac]] ligands can be prepared from [[2,6-Diisopropylaniline|2,6-diisopropylaniline]]. [[Terphenyl]] ligands featuring two diisopropylphenyl substituents, systematically bis(diisopropylphenyl)phenyl, are capable of stabilising high-[[bond order]] compounds such as [[dimetallene]]s.<ref>{{cite journal|last1=Rivard |first1=Eric |last2=Power |first2=Philip P. |title=Multiple Bonding in Heavier Element Compounds Stabilized by Bulky Terphenyl Ligands |journal=Inorganic Chemistry |date=2007 |volume=46 |issue=24 |pages=10047–10064 |doi=10.1021/ic700813h |pmid=17975890 }}</ref> The first stable [[digermyne]] was prepared using DIPP-based terphenyl ligands.<ref>{{cite journal|last1= Stender|first1= Matthias|last2= Phillips|first2= Andrew D.|last3= Wright|first3= Robert J.|last4= Power|first4= Philip P.|title= Synthesis and Characterization of a Digermanium Analogue of an Alkyne|journal= Angewandte Chemie International Edition|date= 2002|volume= 41|issue= 10|pages= 1785–1787|doi= 10.1002/1521-3773(20020517)41:10<1785::AID-ANIE1785>3.0.CO;2-6|pmid= 19750717}}</ref> |
||
Latest revision as of 02:43, 20 November 2025
The 2,6-diisopropylphenyl group, abbreviated Dipp, is an aromatic functional group in organic chemistry. It offers considerable steric bulk, making it of interest for researching unstable compounds and designing ligands in organometallic chemistry.
A major source of diisopropylphenyl substituents is 2,6-diisopropylaniline. Diisopropyl compounds may also be prepared from halobenzene derivatives such as 2,6-diisopropylbromobenzene.[1]

The diisopropylphenyl structure consists of alkane-substituted benzene, so it is not very chemically reactive. It serves instead to provide steric hindrance, limiting reactions of other nearby functional groups.
The diisopropylphenyl structure is found in many ligands calling for steric bulk. NacNac ligands can be prepared from 2,6-diisopropylaniline. Terphenyl ligands featuring two diisopropylphenyl substituents, systematically bis(diisopropylphenyl)phenyl, are capable of stabilising high-bond order compounds such as dimetallenes.[2] The first stable digermyne was prepared using DIPP-based terphenyl ligands.[3]
- ^ Knorr, Rudolf; Ruhdorfer, Jakob; Böhrer, Petra; Bronberger, Hildegard; Räpple, Edith (1994). “Sterically Congested Molecules, 11. The 2,6-Diisopropylphenyl Substituent at C=C and C=N Groups”. Liebigs Annalen der Chemie (4): 433–438. doi:10.1002/jlac.199419940417.
- ^ Rivard, Eric; Power, Philip P. (2007). “Multiple Bonding in Heavier Element Compounds Stabilized by Bulky Terphenyl Ligands”. Inorganic Chemistry. 46 (24): 10047–10064. doi:10.1021/ic700813h. PMID 17975890.
- ^ Stender, Matthias; Phillips, Andrew D.; Wright, Robert J.; Power, Philip P. (2002). “Synthesis and Characterization of a Digermanium Analogue of an Alkyne”. Angewandte Chemie International Edition. 41 (10): 1785–1787. doi:10.1002/1521-3773(20020517)41:10<1785::AID-ANIE1785>3.0.CO;2-6. PMID 19750717.