Tweak.
| ← Previous revision | Revision as of 00:19, 21 January 2026 | ||
| Line 65: | Line 65: | ||
|
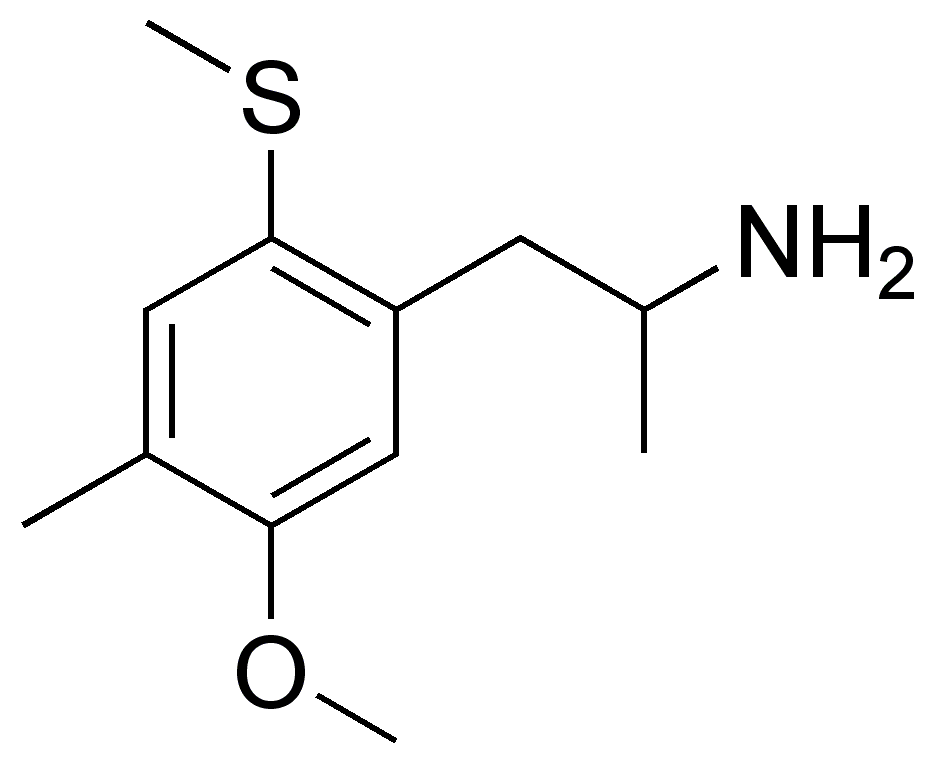
In his book ”[[PiHKAL]]” (”Phenethylamines I Have Known and Loved”) and other publications, [[Alexander Shulgin]] lists 2-TOM’s dose as 60 to 100{{nbsp}}mg [[oral administration|orally]] and its [[duration of action|duration]] as 8 to 10{{nbsp}}hours.<ref name=”PiHKAL” /><ref name=”Shulgin2003″ /> Whereas 2-TOM has a fully effective dose of around 80{{nbsp}}mg, DOM has a fully effective dose of about 5{{nbsp}}mg, and so there is around a 15-fold loss of [[potency (pharmacology)|potency]] with the drug.<ref name=”PiHKAL” /><ref name=”Shulgin2003″ /><ref name=”Nichols1994″ /><ref name=”Marcher-RørstedHalberstadtKlein2020″>{{cite journal | vauthors = Marcher-Rørsted E, Halberstadt AL, Klein AK, Chatha M, Jademyr S, Jensen AA, Kristensen JL | title = Investigation of the 2,5-Dimethoxy Motif in Phenethylamine Serotonin 2A Receptor Agonists | journal = ACS Chem Neurosci | volume = 11 | issue = 9 | pages = 1238–1244 | date = May 2020 | pmid = 32212672 | doi = 10.1021/acschemneuro.0c00129 | url = | quote = Shulgin observed that replacement of either the 2-position (10, Figure 2) or the 5- position (11, Figure 2) oxygen in 9 with a sulfur atom reduces its hallucinogenic potency by approximately 15- or 10-fold, respectively.13 Replacing both oxygen atoms with sulfur (12, Figure 2) completely abolished activity.}}</ref> In addition, it has a shorter duration than DOM, with DOM having a listed duration of 14 to 20{{nbsp}}hours.<ref name=”PiHKAL” /> The effects of 2-TOM have been reported to include [[closed-eye visual]]s, feeling strange, “superb body feeling”, pleasantness, bodily awareness, and feeling heavy.<ref name=”PiHKAL” /> It has none of the neurological or physical “roughness” that was observed with 5-TOM.<ref name=”PiHKAL” />
|
In his book ”[[PiHKAL]]” (”Phenethylamines I Have Known and Loved”) and other publications, [[Alexander Shulgin]] lists 2-TOM’s dose as 60 to 100{{nbsp}}mg [[oral administration|orally]] and its [[duration of action|duration]] as 8 to 10{{nbsp}}hours.<ref name=”PiHKAL” /><ref name=”Shulgin2003″ /> Whereas 2-TOM has a fully effective dose of around 80{{nbsp}}mg, DOM has a fully effective dose of about 5{{nbsp}}mg, and so there is around a 15-fold loss of [[potency (pharmacology)|potency]] with the drug.<ref name=”PiHKAL” /><ref name=”Shulgin2003″ /><ref name=”Nichols1994″ /><ref name=”Marcher-RørstedHalberstadtKlein2020″>{{cite journal | vauthors = Marcher-Rørsted E, Halberstadt AL, Klein AK, Chatha M, Jademyr S, Jensen AA, Kristensen JL | title = Investigation of the 2,5-Dimethoxy Motif in Phenethylamine Serotonin 2A Receptor Agonists | journal = ACS Chem Neurosci | volume = 11 | issue = 9 | pages = 1238–1244 | date = May 2020 | pmid = 32212672 | doi = 10.1021/acschemneuro.0c00129 | url = | quote = Shulgin observed that replacement of either the 2-position (10, Figure 2) or the 5- position (11, Figure 2) oxygen in 9 with a sulfur atom reduces its hallucinogenic potency by approximately 15- or 10-fold, respectively.13 Replacing both oxygen atoms with sulfur (12, Figure 2) completely abolished activity.}}</ref> In addition, it has a shorter duration than DOM, with DOM having a listed duration of 14 to 20{{nbsp}}hours.<ref name=”PiHKAL” /> The effects of 2-TOM have been reported to include [[closed-eye visual]]s, feeling strange, “superb body feeling”, pleasantness, bodily awareness, and feeling heavy.<ref name=”PiHKAL” /> It has none of the neurological or physical “roughness” that was observed with 5-TOM.<ref name=”PiHKAL” />
|
||
|
The [[chemical synthesis]] of 2-TOM has been described.<ref name=”PiHKAL” /><ref name=”JacobShulgin1983″ /> The [[substituted phenethylamine|phenethylamine]] [[structural analog|analogue]], [[2C-2-TOM]] (2-thio-2C-D), has been synthesized, but was not tested and its properties are unknown.<ref name=”PiHKAL” /> [[Bis-TOM]], the 2,5-dimethylthio analogue of DOM, was synthesized and tested, but was inactive at doses of up to 160{{nbsp}}mg or approximately 50{{nbsp}}times the minimum effective dose of DOM.<ref name=”PiHKAL” /><ref name=”Shulgin2003″ /><ref name=”Marcher-RørstedHalberstadtKlein2020″ /><ref name=”JacobShulgin1983″ />
|
The [[chemical synthesis]] of 2-TOM has been described.<ref name=”PiHKAL” /><ref name=”JacobShulgin1983″ /> The [[substituted phenethylamine|phenethylamine]] [[structural analog|analogue]], [[2C-2-TOM]] (2-thio-2C-D), has been synthesized, but was not tested and its properties are unknown.<ref name=”PiHKAL” /> [[Bis-TOM]], the 2,5-dimethylthio analogue of DOM, was synthesized and tested, but was inactive at doses of up to 160{{nbsp}}mg or approximately 50{{nbsp}}times the minimum effective dose of DOM.<ref name=”PiHKAL” /><ref name=”Shulgin2003″ /><ref name=”Marcher-RørstedHalberstadtKlein2020″ /><ref name=”JacobShulgin1983″ />
|
||
|
2-TOM was first described in the [[scientific literature]] by [[Alexander Shulgin]] and [[Peyton Jacob III]] in 1983.<ref name=”JacobShulgin1983″>{{cite journal | vauthors = Jacob P, Shulgin AT | title = Sulfur analogues of psychotomimetic agents. 2. Analogues of (2,5-dimethoxy-4-methylphenyl)-and (2,5-dimethoxy-4-ethylphenyl)isopropylamine | journal = J Med Chem | volume = 26 | issue = 5 | pages = 746–752 | date = May 1983 | pmid = 6842515 | doi = 10.1021/jm00359a021 | url = }}</ref> Subsequently, it was described in greater detail by Shulgin in ”PiHKAL” in 1991.<ref name=”PiHKAL” />
|
2-TOM was first described in the [[scientific literature]] by [[Alexander Shulgin]] and [[Peyton Jacob III]] in 1983.<ref name=”JacobShulgin1983″>{{cite journal | vauthors = Jacob P, Shulgin AT | title = Sulfur analogues of psychotomimetic agents. 2. Analogues of (2,5-dimethoxy-4-methylphenyl)-and (2,5-dimethoxy-4-ethylphenyl)isopropylamine | journal = J Med Chem | volume = 26 | issue = 5 | pages = 746–752 | date = May 1983 | pmid = 6842515 | doi = 10.1021/jm00359a021 | url = }}</ref> Subsequently, it was described in greater detail by Shulgin in ”PiHKAL” in 1991.<ref name=”PiHKAL” />
|
||