[[File:Absorption spectrum NOCl.png|thumb|center|upright=2.0|The absorption spectrum for the NOCl molecule on excitation to the S1 state]]
[[File:Absorption spectrum NOCl.png|thumb|center|upright=2.0|The absorption spectrum for the NOCl molecule on excitation to the S1 state]]
==Further Reading==
* {{cite journal | last1=Meyer | first1=H.-D. | last2=Manthe | first2=U. | last3=Cederbaum | first3=L.S. | title=The multi-configurational time-dependent Hartree approach | journal=Chemical Physics Letters | publisher=Elsevier BV | volume=165 | issue=1 | year=1990 | issn=0009-2614 | doi=10.1016/0009-2614(90)87014-i | pages=73–78| bibcode=1990CPL…165…73M }}
* {{cite journal | last1=Manthe | first1=U. | last2=Meyer | first2=H.‐D. | last3=Cederbaum | first3=L. S. | title=Wave‐packet dynamics within the multiconfiguration Hartree framework: General aspects and application to NOCl | journal=The Journal of Chemical Physics | publisher=AIP Publishing | volume=97 | issue=5 | year=1992 | issn=0021-9606 | doi=10.1063/1.463007 | pages=3199–3213| bibcode=1992JChPh..97.3199M }}
*{{cite journal | last1=Beck | first1=M. H. |last2=Jäckle|first2=A.|last3=Worth|first3=G. A.|last4=Meyer|first4=H.-D.| title=The multiconfiguration time-dependent Hartree (MCTDH) method: a highly efficient algorithm for propagating wavepackets | journal=Physics Reports | publisher=Elsevier BV | volume=324 | issue=1 | year=2000 | issn=0370-1573 | doi=10.1016/s0370-1573(99)00047-2 | pages=1–105| bibcode=2000PhR…324….1B }}
==External links==
*[http://mctdh.uni-hd.de The Heidelberg MCTDH Homepage]
[[Category:Quantum chemistry]]
[[Category:Quantum chemistry]]
[[Category:Scattering]]
[[Category:Scattering]]
{{quantum-chemistry-stub}}
{{quantum-chemistry-stub}}
{{scattering-stub}}
{{scattering-stub}}
==References==
Multi-configuration time-dependent Hartree (MCTDH) is a general algorithm to solve the time-dependent Schrödinger equation for multidimensional dynamical systems consisting of distinguishable particles.
MCTDH can thus determine the quantal motion of the nuclei of a molecular system evolving on one or several coupled electronic potential energy surfaces. It is an approximate method whose numerical efficiency decreases with growing accuracy [1].
MCTDH is suited for multi-dimensional problems, in particular for problems that are difficult or even impossible to solve in conventional ways [citation needed].
Where the number of configurations is given by the product . The single particle functions (SPFs), , are expressed in a time-independent basis set:
Where is a primative basis function, in general a Discrete Variable
Representation (DVR) that is dependent on coordinate [2]. If , one returns to the Time Dependent Hartree (TDH) approach[3]. In MCTDH, both the coefficients and the basis function are time-dependent and optimized using the variational principle.
Equations of Motion
[edit]
Lagrangian Variational Principle
[edit]
Where:
Which is subject to the boundary conditions . After integration, one obtains:
McLachlan Variational Principle
[edit]
Where only the time derivative is to be varied. We can rewrite this norm squared term as a scalar product, and vary the bra and ket side of the product:
Dirac-Frenkel Variational Principle
[edit]
If each variation of is an allowed variation, then both the the Lagrangian and the McLanchlan Variational Principal turn into the Dirac-Frenkel Variational Principle:
Which simplest and thus preferred method of deriving the equations of motion[2].
The original ansatz of MCTDH generates a single layer tensor tree; however, there is a limit to the size and complexity this single layer can handle. This prompted the development of a multilayer (ML)-MCTDH ansatz by Manthe[4] which was then generalized by Vendrell and Meyer[5].
Tensor Tree Formalism
[edit]
Multiple layers are generated through the creation of a tensor tree of nodes linking the modes (DOFs). Solving the tree layout is an NP-hard problem, but strides have been taken to automate this process through mode correlations by Mendive-Tapia[6].

The generalized ML expansion of Meyer[5] can be written as follows:
Where the coordinates are combined as
Equations of motion
[edit]
Where the equations of motion are now represented as follows:
The SPF EOMs are formally defined the same for all layers:
Where is a Hermitian gauge operator defined as follows:
Examples of Uses in Literature
[edit]
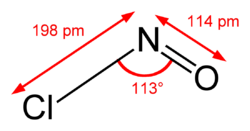
The first verification of the MCTDH method was with the NOCl molecule. It’s size and asymmetry makes it a perfect test bed for MCTDH: it is small and simple enough for its numerics to be manually verified, yet complicated enough for it to already squeeze advantages against conventional product-basis methods.[7]

The solvation of the hydronium ion is a topic of continued research. Researchers have been able to successfully use MCTDH to model the Zundel [8] and Eigen[9] ions in close agreement with experiment.
| Method | Degrees of Freedom Possible |
|---|---|
| Conventional Methods (e.g. TDH) | 6 |
| MCTDH | 12 [1] |
| ML-MCTDH | 24+ [5] |
| ML-MCTDH with the Spin-Boson Model | 1000+[10] |
For a typical input in ML-MCTDH to be run, a node tree, potential energy surface, and equations of motion must be generated by the user [11]. These prerequisites—along with total compute time—soft-cap the size of systems able to be studied with ML-MCTDH; however, advances in neural networks have been shown to address the difficulty of the generation of potential energy surfaces [12]. These issues can also by circumvented by using the spin-boson or other similar bath models that do not pose the same assignment challenges [10].
Software Packages Implementing the MCTDH Method
[edit]
Example Usage of the Heidelberg Package for NOCl
[edit]
Input and Operator File
[edit]
| nocl0.inp | nocl0.op |
|---|---|
RUN-SECTION
relaxation
tfinal= 50.0
tout= 10.0
name = nocl0
overwrite
output psi=double timing
end-run-section
OPERATOR-SECTION
opname = nocl0
end-operator-section
SBASIS-SECTION
rd = 5
rv = 5
theta = 5
end-sbasis-section
pbasis-section
#Label DVR N Parameter
rd sin 36 3.800 5.600
rv HO 24 2.136 0.272,ev 13615.5
theta Leg 60 0 0
end-pbasis-section
INTEGRATOR-SECTION
CMF/var = 0.50 , 1.0d-5
BS/spf = 10 , 1.0d-7
SIL/A = 12 , 1.0d-7
end-integrator-section
INIT_WF-SECTION
build
rd gauss 4.315 0.0 0.0794
rv HO 2.151 0.0 0.218,eV 13615.5
theta gauss 2.22 0.0 0.0745
end-build
end-init_wf-section
ALLOC-SECTION
maxkoe=160
maxhtm=220
maxhop=220
maxsub=60
maxLMR=1
maxdef=85
maxedim=1
maxfac=25
maxmuld=1
maxnhtmshift=1
end-alloc-section
end-input
|
OP_DEFINE-SECTION
title
NOCl S0 surface
end-title
end-op_define-section
PARAMETER-SECTION
mass_rd = 16.1538, AMU
mass_rv = 7.4667, AMU
end-parameter-section
HAMILTONIAN-SECTION
---------------------------------------------------------
modes | rd | rv | theta
---------------------------------------------------------
0.5/mass_rd | q^-2 | 1 | j^2
0.5/mass_rv | 1 | q^-2 | j^2
1.0 | KE | 1 | 1
1.0 | 1 | KE | 1
1.0 |1&2&3 V
---------------------------------------------------------
end-hamiltonian-section
LABELS-SECTION
V = srffile {nocl0um, default}
end-labels-section
end-operator
|
Output Absorption Spectrum
[edit]

- ^ a b Meyer, Hans-Dieter. “Multi-Configurarion time-dependent Hartree”. Theoretical Chemistry Group Heidelberg. Heidelberg University. Retrieved 25 October 2025.
- ^ a b Meyer, Hans-Dieter. “Introduction to MCTDH” (PDF). Theoretical Chemistry Group. Heidelberg University. Retrieved 25 October 2025.
- ^ McLachlan, A. D.; Ball, M. A. (1964). “Time-Dependent Hartree—Fock Theory for Molecules”. Reviews of Modern Physics. 36 (3): 844–855. doi:10.1103/RevModPhys.36.844. Retrieved 25 October 2025.
- ^ Manthe, Uwe (2008). “A multilayer multiconfigurational time-dependent Hartree approach for quantum dynamics on general potential energy surfaces”. The Journal of Chemical Physics. 128 (16): 164116. doi:10.1063/1.2902982. Retrieved 25 October 2025.
- ^ a b c Vendrell, Oriol; Meyer, Hans-Dieter (2011). “Multilayer multiconfiguration time-dependent Hartree method: Implementation and applications to a Henon–Heiles Hamiltonian and to pyrazine”. The Journal of Chemical Physics. 134 (4): 044135. doi:10.1063/1.3535541. Retrieved 25 October 2025.
- ^ Mendive-Tapia, David; Meyer, Hans-Dieter; Vendrell, Oriol (2023). “Optimal Mode Combination in the Multiconfiguration Time-Dependent Hartree Method through Multivariate Statistics: Factor Analysis and Hierarchical Clustering”. Journal of Chemical Theory and Computation. 19 (4): 1144–1156. doi:10.1021/acs.jctc.2c01089.
- ^ Manthe, Uwe; Meyer, Hans-Dieter; Cederbaum, Lorenz (1992). “Wave‐packet dynamics within the multiconfiguration Hartree framework: General aspects and application to NOCl”. The Journal of Chemical Physics. 97 (5): 3199–3213. doi:10.1063/1.463007. Retrieved 27 October 2025.
- ^ Vendrell, Oriol; Gatti, Fabien; Meyer, Hans-Dieter (2007). “Full dimensional (15-dimensional) quantum-dynamical simulation of the protonated water dimer. II. Infrared spectrum and vibrational dynamics”. The Journal of Chemical Physics. 127 (18): 184303. doi:10.1063/1.2787596. Retrieved 25 October 2025.
- ^ Schröder, Markus; Gatti, Fabien; Lauvergnat, David; Meyer, Hans-Dieter; Vendrell, Oriol (2022). “The coupling of the hydrated proton to its first solvation shell”. Nature Communications. 13: 6170. doi:10.1038/s41467-022-33650-w. Retrieved 25 October 2025.
- ^ a b Wang, Haobin (2019). “Quantum Phase Transition in the Spin-Boson Model: A Multilayer Multiconfiguration Time-Dependent Hartree Study”. The Journal of Physical Chemistry A. 123 (9): 1882–1893. doi:10.1021/acs.jpca.8b11136. Retrieved 25 October 2025.
- ^ Meyer, Hans-Dieter. “The Heidelberg MCTDH Package: A set of programs for multi-dimensional quantum dynamics” (PDF). User’s Guide. Heidelberg University. Retrieved 25 October 2025.
- ^ Marx, Dominik. “RubNNet4MD”. Center for Theoretical Chemistry. Ruhr-Universität Bochum. Retrieved 25 October 2025.























