=== Type IVB secretion system ===
=== Type IVB secretion system ===
The bacteria use a [[Type IV secretion system|type IVB secretion system]] known as Icm/Dot (intracellular multiplication / defect in organelle trafficking genes) to inject over 100 effector proteins into the host.<ref>{{Cite journal |last1=van Schaik |first1=Erin J. |last2=Chen |first2=Chen |last3=Mertens |first3=Katja |last4=Weber |first4=Mary M. |last5=Samuel |first5=James E. |date=August 2013 |title=Molecular pathogenesis of the obligate intracellular bacterium Coxiella burnetii |journal=Nature Reviews. Microbiology |volume=11 |issue=8 |pages=561–573 |doi=10.1038/nrmicro3049 |issn=1740-1534 |pmc=4134018 |pmid=23797173}}</ref><ref>{{Cite journal |last1=Newton |first1=Hayley J. |last2=Kohler |first2=Lara J. |last3=McDonough |first3=Justin A. |last4=Temoche-Diaz |first4=Morayma |last5=Crabill |first5=Emerson |last6=Hartland |first6=Elizabeth L. |last7=Roy |first7=Craig R. |date=July 2014 |title=A screen of Coxiella burnetii mutants reveals important roles for Dot/Icm effectors and host autophagy in vacuole biogenesis |journal=PLOS Pathogens |volume=10 |issue=7 |article-number=e1004286 |doi=10.1371/journal.ppat.1004286 |doi-access=free |issn=1553-7374 |pmc=4117601 |pmid=25080348}}</ref> These effectors increase the bacterium’s ability to survive and replicate within host cells by modulating multiple host pathways, including blocking apoptosis, inhibiting immune responses, and altering vesicle trafficking.<ref name=”:16″ /><ref>{{Cite journal |last1=Lührmann |first1=Anja |last2=Nogueira |first2=Catarina V. |last3=Carey |first3=Kimberly L. |last4=Roy |first4=Craig R. |date=2010-11-02 |title=Inhibition of pathogen-induced apoptosis by a Coxiella burnetii type IV effector protein |journal=Proceedings of the National Academy of Sciences of the United States of America |volume=107 |issue=44 |pages=18997–19001 |doi=10.1073/pnas.1004380107 |doi-access=free |issn=1091-6490 |pmc=2973885 |pmid=20944063 |bibcode=2010PNAS..10718997L }}</ref> In ”[[Legionella pneumophila]]”, a related [[Gammaproteobacteria|Gammaproteobacterium]] that uses the same secretion system, these effectors enhance survival by preventing fusion of the bacteria-containing vacuole with degradative [[Endosome|endosomes.]]<ref>{{Cite journal |last1=Pan |first1=Xiaoxiao |last2=Lührmann |first2=Anja |last3=Satoh |first3=Ayano |last4=Laskowski-Arce |first4=Michelle A. |last5=Roy |first5=Craig R. |date=2008-06-20 |title=Ankyrin repeat proteins comprise a diverse family of bacterial type IV effectors |journal=Science (New York, N.Y.) |volume=320 |issue=5883 |pages=1651–1654 |doi=10.1126/science.1158160 |issn=1095-9203 |pmc=2514061 |pmid=18566289 |bibcode=2008Sci…320.1651P }}</ref>
The bacteria use a [[Type IV secretion system|type IVB secretion system]] known as Icm/Dot (intracellular multiplication / defect in organelle trafficking genes) to inject over 100 effector proteins into the host.<ref>{{Cite journal |last1=van Schaik |first1=Erin J. |last2=Chen |first2=Chen |last3=Mertens |first3=Katja |last4=Weber |first4=Mary M. |last5=Samuel |first5=James E. |date=August 2013 |title=Molecular pathogenesis of the obligate intracellular bacterium Coxiella burnetii |journal=Nature Reviews. Microbiology |volume=11 |issue=8 |pages=561–573 |doi=10.1038/nrmicro3049 |issn=1740-1534 |pmc=4134018 |pmid=23797173}}</ref><ref>{{Cite journal |last1=Newton |first1=Hayley J. |last2=Kohler |first2=Lara J. |last3=McDonough |first3=Justin A. |last4=Temoche-Diaz |first4=Morayma |last5=Crabill |first5=Emerson |last6=Hartland |first6=Elizabeth L. |last7=Roy |first7=Craig R. |date=July 2014 |title=A screen of Coxiella burnetii mutants reveals important roles for Dot/Icm effectors and host autophagy in vacuole biogenesis |journal=PLOS Pathogens |volume=10 |issue=7 |article-number=e1004286 |doi=10.1371/journal.ppat.1004286 |doi-access=free |issn=1553-7374 |pmc=4117601 |pmid=25080348}}</ref> These effectors increase the bacterium’s ability to survive and replicate within host cells by modulating multiple host pathways, including blocking apoptosis, inhibiting immune responses, and altering vesicle trafficking.<ref name=”:16″ /><ref>{{Cite journal |last1=Lührmann |first1=Anja |last2=Nogueira |first2=Catarina V. |last3=Carey |first3=Kimberly L. |last4=Roy |first4=Craig R. |date=2010-11-02 |title=Inhibition of pathogen-induced apoptosis by a Coxiella burnetii type IV effector protein |journal=Proceedings of the National Academy of Sciences of the United States of America |volume=107 |issue=44 |pages=18997–19001 |doi=10.1073/pnas.1004380107 |doi-access=free |issn=1091-6490 |pmc=2973885 |pmid=20944063 |bibcode=2010PNAS..10718997L }}</ref> In ”[[Legionella pneumophila]]”, a related [[Gammaproteobacteria|Gammaproteobacterium]] that uses the same secretion system, these effectors enhance survival by preventing fusion of the bacteria-containing vacuole with degradative [[Endosome|endosomes.]]<ref>{{Cite journal |last1=Pan |first1=Xiaoxiao |last2=Lührmann |first2=Anja |last3=Satoh |first3=Ayano |last4=Laskowski-Arce |first4=Michelle A. |last5=Roy |first5=Craig R. |date=2008-06-20 |title=Ankyrin repeat proteins comprise a diverse family of bacterial type IV effectors |journal=Science (New York, N.Y.) |volume=320 |issue=5883 |pages=1651–1654 |doi=10.1126/science.1158160 |issn=1095-9203 |pmc=2514061 |pmid=18566289 |bibcode=2008Sci…320.1651P }}</ref>
=== Treatment and prevention ===
=== Treatment and prevention ===
Acute Q fever is typically treated with [[doxycycline]], which shortens illness duration and reduces the risk of progression to chronic disease. Other antibiotics, including [[Macrolide|macrolides]], [[Trimethoprim/sulfamethoxazole|co-trimoxazole]], [[Quinolone antibiotic|quinolones]], and [[Β-Lactam|beta-lactams]], have been used but show less consistent effectiveness. [[Serology|Serologic]] tests may not become positive until 1–2 weeks after exposure, so the CDC recommends initiating doxycycline based on clinical suspicion rather than waiting for laboratory confirmation.<ref name=”:3″>{{Cite journal |last=Kersh |first=Gilbert J. |date=2013-11 |title=Antimicrobial therapies for Q fever |url=https://pubmed.ncbi.nlm.nih.gov/24073941 |journal=Expert Review of Anti-Infective Therapy |volume=11 |issue=11 |pages=1207–1214 |doi=10.1586/14787210.2013.840534 |issn=1744-8336 |pmc=4608426 |pmid=24073941}}</ref><ref>{{Cite web |last=CDC |date=2025-05-21 |title=Clinical Guidance for Q fever |url=https://www.cdc.gov/q-fever/hcp/clinical-guidance/index.html |access-date=2025-11-19 |website=Q Fever |language=en-us}}</ref>
Acute Q fever is typically treated with [[doxycycline]], which shortens illness duration and reduces the risk of progression to chronic disease. Other antibiotics, including [[Macrolide|macrolides]], [[Trimethoprim/sulfamethoxazole|co-trimoxazole]], [[Quinolone antibiotic|quinolones]], and [[Β-Lactam|beta-lactams]], have been used but show less consistent effectiveness. [[Serology|Serologic]] tests may not become positive until 1–2 weeks after exposure, so the CDC recommends initiating doxycycline based on clinical suspicion rather than waiting for laboratory confirmation.<ref name=”:3″>{{Cite journal |last=Kersh |first=Gilbert J. |date=2013 |title=Antimicrobial therapies for Q fever |url=https://pubmed.ncbi.nlm.nih.gov/24073941 |journal=Expert Review of Anti-Infective Therapy |volume=11 |issue=11 |pages=1207–1214 |doi=10.1586/14787210.2013.840534 |issn=1744-8336 |pmc=4608426 |pmid=24073941}}</ref><ref>{{Cite web |last=CDC |date=2025-05-21 |title=Clinical Guidance for Q fever |url=https://www.cdc.gov/q-fever/hcp/clinical-guidance/index.html |access-date=2025-11-19 |website=Q Fever |language=en-us}}</ref>
Chronic Q fever, particularly [[endocarditis]], requires prolonged combination therapy. The most effective regimen is doxycycline with [[hydroxychloroquine]] for at least 18 months, and patients undergoing treatment require long-term serologic monitoring to ensure clearance of infection and detect relapse.<ref name=”:3″ />
Chronic Q fever, particularly [[endocarditis]], requires prolonged combination therapy. The most effective regimen is doxycycline with [[hydroxychloroquine]] for at least 18 months, and patients undergoing treatment require long-term serologic monitoring to ensure clearance of infection and detect relapse.<ref name=”:3″ />
== Prevalence and host ==
== Prevalence and host ==
”Coxiella” ”burnetii” is a globally distributed microbe (excluding New Zealand and Antarctica) found commonly in domestic reservoirs such as sheep, goats, and cattle.<ref name=”:7″ /> The microbe is spread to other organisms through the inhalation of contaminated [[Aerosol|aerosols]]. It presents asymptomatically in animals, and proves to cause chronic disease in nearby humans. The microbe is spread from domestic animals to human hosts during [[parturition]] or slaughter of the domestic animal.<ref name=”:1″ /> Ticks can spread the microbe across generations of livestock through [[transovarial transmission]]. Other insects such as flies, cockroaches, and lice can also act as reservoirs.<ref name=”:7″>{{Cite journal |last1=Selim |first1=Abdelfattah |last2=Marzok |first2=Mohamed |last3=Gattan |first3=Hattan S. |last4=Ismail |first4=Hesham |date=2025-10-07 |title=Risk factors influencing Coxiella burnetii seropositivity in water Buffalo (Bubalus bubalis) populations of Egypt’s Nile Delta |journal=PLOS ONE |language=en |volume=20 |issue=10 |article-number=e0333680 |doi=10.1371/journal.pone.0333680 |doi-access=free |issn=1932-6203 |pmc=12503306 |pmid=41056260 |bibcode=2025PLoSO..2033680S }}</ref>
”Coxiella” ”burnetii” is a globally distributed microbe (excluding New Zealand and Antarctica) found commonly in domestic reservoirs such as sheep, goats, and cattle.<ref name=”:7″ /> The microbe is spread to other organisms through the inhalation of contaminated [[Aerosol|aerosols]]. It presents asymptomatically in animals, and proves to cause chronic disease in nearby humans. The microbe is spread from domestic animals to human hosts during [[parturition]] or slaughter of the domestic animal.<ref name=”:1″ /> Ticks can spread the microbe across generations of livestock through [[transovarial transmission]]. Other insects such as flies, cockroaches, and lice can also act as reservoirs.<ref name=”:7″>{{Cite journal |last1=Selim |first1=Abdelfattah |last2=Marzok |first2=Mohamed |last3=Gattan |first3=Hattan S. |last4=Ismail |first4=Hesham |date=2025-10-07 |title=Risk factors influencing Coxiella burnetii seropositivity in water Buffalo (Bubalus bubalis) populations of Egypt’s Nile Delta |journal=PLOS |language=en |volume=20 |issue=10 |article-number=e0333680 |doi=10.1371/journal.pone.0333680 |doi-access=free |issn=1932-6203 |pmc=12503306 |pmid=41056260 |bibcode=2025PLoSO..2033680S }}</ref>
[[Q fever]] is present in German human populations with 27-100 cases reported annually. It is estimated to affect 50 per every 100,000 inhabitants annually in France. The UK has faced 904 cases between 2000 and 2015, as well as major outbreaks in 2002 and 2007. Italy has experienced two different outbreaks of human infection in [[Como]] prison and [[Vicenza]] respectively. Spain has a high prevalence of ”C. burnetii” infection in human populations in [[Basque Country (autonomous community)|Basque]] and [[Navarre]]. The annual reported cases of human ”C. burnetii” infection in the US went up from 19 cases in 2000 to around 160-180 cases per year, with 178 acute cases in 2019. In the US, cases of human infection were mainly found in the West or the Great Plains regions of the country. Canada has a low human incidence of infection. In Quebec, it was reported that there were 0.4 cases per 100,000 inhabitants in 2017. In Alberta, only 39 cases were reported.<ref name=”:1″>{{Cite journal |last1=Rahal |first1=Mohamed |last2=Salhi |first2=Omar |last3=Ouchetati |first3=Imane |last4=Khelifi Touhami |first4=Nadjet Amina |last5=Ouchene |first5=Nassim |date=2025-11-01 |title=Global epidemiology and molecular typing of Coxiella burnetii: A systematic review of Q fever in humans and animals |url=https://www.sciencedirect.com/science/article/pii/S0147957125001092 |journal=Comparative Immunology, Microbiology and Infectious Diseases |volume=123 |article-number=102401 |doi=10.1016/j.cimid.2025.102401 |pmid=40902447 |issn=0147-9571|url-access=subscription }}</ref> ”Coxiella” ”burnetii” has been observed with a greater prevalence in populations in close proximity to animals as well as homeless populations in Brazil.<ref>{{Cite journal |last1=de França |first1=Danilo Alves |last2=Kmetiuk |first2=Louise Bach |last3=do Couto |first3=Anahi Chechia |last4=Langoni |first4=Helio |last5=Biondo |first5=Alexander Welker |date=2025-08-03 |title=Coxiella burnetii and HIV infection in people experiencing homelessness |journal=Scientific Reports |language=en |volume=15 |issue=1 |article-number=28312 |doi=10.1038/s41598-025-09422-z |issn=2045-2322 |pmc=12319078 |pmid=40754629 |bibcode=2025NatSR..1528312D }}</ref> In Algeria, residents of rural areas were at a three times higher risk for ”Coxiella” ”burnetii” infections compared to inhabitants of urban areas.<ref name=”:17″>{{Cite journal |last1=Lacheheb |first1=A. |last2=Raoult |first2=D. |date=2009-12-01 |title=Seroprevalence of Q-fever in Algeria |url=https://www.clinicalmicrobiologyandinfection.org/article/S1198-743X(14)63530-8/fulltext |journal=Clinical Microbiology and Infection |language=English |volume=15 |pages=167–168 |doi=10.1111/j.1469-0691.2008.02211.x |issn=1198-743X |pmid=19793121}}</ref> [[Isiolo County]], Kenya has been found to have a 44.7% [[seroprevalence]] of ”Coxiella burnetii” in humans.<ref name=”:8″>{{Cite journal |last1=Mutisya |first1=Wilfred Mutuku |last2=Akoko |first2=James M. |last3=Mwatondo |first3=Athman |last4=Muturi |first4=Mathew |last5=Nthiwa |first5=Daniel |last6=Abkallo |first6=Hussein M. |last7=Nyamota |first7=Richard |last8=Wachira |first8=Timothy |last9=Gathura |first9=Peter |last10=Bett |first10=Bernard |date=2025-10-17 |title=Sero-epidemiology of Coxiella burnetii in livestock and humans in Isiolo county Kenya |journal=PLOS Neglected Tropical Diseases |language=en |volume=19 |issue=10 |article-number=e0013557 |doi=10.1371/journal.pntd.0013557 |doi-access=free |issn=1935-2735 |pmc=12551958 |pmid=41105740}}</ref> In [[Kwara State]], Nigeria, ”C. burnetii” was found with a 18.8% prevalence in milk and cheese, contributing to further transmission of Q fever through consumption of milk products.<ref>{{Cite journal |last1=Elelu |first1=Nusirat |last2=Chinedu |first2=Nwachukwu Raymond |last3=Yakub-Obalowu |first3=Balkees A. |last4=Odetokun |first4=Ismail A. |last5=Al-Mustapha |first5=Ahmad I. |date=2025-01-07 |title=Real-time PCR detection of Coxiella burnetii in dairy products in Kwara State, Nigeria: a public health concern |journal=BMC Veterinary Research |volume=21 |issue=1 |page=8 |doi=10.1186/s12917-024-04449-2 |doi-access=free |issn=1746-6148 |pmc=11706007 |pmid=39773743}}</ref>
[[Q fever]] is present in German human populations with 27-100 cases reported annually. It is estimated to affect 50 per every 100,000 inhabitants annually in France. The UK has faced 904 cases between 2000 and 2015, as well as major outbreaks in 2002 and 2007. Italy has experienced two different outbreaks of human infection in [[Como]] prison and [[Vicenza]] respectively. Spain has a high prevalence of ”C. burnetii” infection in human populations in [[Basque Country (autonomous community)|Basque]] and [[Navarre]]. The annual reported cases of human ”C. burnetii” infection in the US went up from 19 cases in 2000 to around 160-180 cases per year, with 178 acute cases in 2019. In the US, cases of human infection were mainly found in the West or the Great Plains regions of the country. Canada has a low human incidence of infection. In Quebec, it was reported that there were 0.4 cases per 100,000 inhabitants in 2017. In Alberta, only 39 cases were reported.<ref name=”:1″>{{Cite journal |last1=Rahal |first1=Mohamed |last2=Salhi |first2=Omar |last3=Ouchetati |first3=Imane |last4=Khelifi Touhami |first4=Nadjet Amina |last5=Ouchene |first5=Nassim |date=2025-11-01 |title=Global epidemiology and molecular typing of Coxiella burnetii: A systematic review of Q fever in humans and animals |url=https://www.sciencedirect.com/science/article/pii/S0147957125001092 |journal=Comparative Immunology, Microbiology and Infectious Diseases |volume=123 |article-number=102401 |doi=10.1016/j.cimid.2025.102401 |pmid=40902447 |issn=0147-9571|url-access=subscription }}</ref> ”Coxiella” ”burnetii” has been observed with a greater prevalence in populations in close proximity to animals as well as homeless populations in Brazil.<ref>{{Cite journal |last1=de França |first1=Danilo Alves |last2=Kmetiuk |first2=Louise Bach |last3=do Couto |first3=Anahi Chechia |last4=Langoni |first4=Helio |last5=Biondo |first5=Alexander Welker |date=2025-08-03 |title=Coxiella burnetii and HIV infection in people experiencing homelessness |journal=Scientific Reports |language=en |volume=15 |issue=1 |article-number=28312 |doi=10.1038/s41598-025-09422-z |issn=2045-2322 |pmc=12319078 |pmid=40754629 |bibcode=2025NatSR..1528312D }}</ref> In Algeria, residents of rural areas were at a three times higher risk for ”Coxiella” ”burnetii” infections compared to inhabitants of urban areas.<ref name=”:17″>{{Cite journal |last1=Lacheheb |first1=A. |last2=Raoult |first2=D. |date=2009-12-01 |title=Seroprevalence of Q-fever in Algeria |url=https://www.clinicalmicrobiologyandinfection.org/article/S1198-743X(14)63530-8/fulltext |journal=Clinical Microbiology and Infection |language=English |volume=15 |pages=167–168 |doi=10.1111/j.1469-0691.2008.02211.x |issn=1198-743X |pmid=19793121}}</ref> [[Isiolo County]], Kenya has been found to have a 44.7% [[seroprevalence]] of ”Coxiella burnetii” in humans.<ref name=”:8″>{{Cite journal |last1=Mutisya |first1=Wilfred Mutuku |last2=Akoko |first2=James M. |last3=Mwatondo |first3=Athman |last4=Muturi |first4=Mathew |last5=Nthiwa |first5=Daniel |last6=Abkallo |first6=Hussein M. |last7=Nyamota |first7=Richard |last8=Wachira |first8=Timothy |last9=Gathura |first9=Peter |last10=Bett |first10=Bernard |date=2025-10-17 |title=Sero-epidemiology of Coxiella burnetii in livestock and humans in Isiolo county Kenya |journal=PLOS Neglected Tropical Diseases |language=en |volume=19 |issue=10 |article-number=e0013557 |doi=10.1371/journal.pntd.0013557 |doi-access=free |issn=1935-2735 |pmc=12551958 |pmid=41105740}}</ref> In [[Kwara State]], Nigeria, ”C. burnetii” was found with a 18.8% prevalence in milk and cheese, contributing to further transmission of Q fever through consumption of milk products.<ref>{{Cite journal |last1=Elelu |first1=Nusirat |last2=Chinedu |first2=Nwachukwu Raymond |last3=Yakub-Obalowu |first3=Balkees A. |last4=Odetokun |first4=Ismail A. |last5=Al-Mustapha |first5=Ahmad I. |date=2025-01-07 |title=Real-time PCR detection of Coxiella burnetii in dairy products in Kwara State, Nigeria: a public health concern |journal=BMC Veterinary Research |volume=21 |issue=1 |page=8 |doi=10.1186/s12917-024-04449-2 |doi-access=free |issn=1746-6148 |pmc=11706007 |pmid=39773743}}</ref>
The United States ended its [[biological warfare]] program in 1969. When it did, ”C. burnetii” was one of seven agents it had standardized as biological weapons.<ref name=croddyhart>{{cite book |last1=Croddy |first1=Eric C. |last2=Hart |first2=C. Perez-Armendariz J. |title=Chemical and Biological Warfare |url=https://books.google.com/books?id=fGp53P6ijqsC&pg=PA30 |publisher=Springer |year=2002 |pages=30–31 |isbn=0-387-95076-1 }}</ref> There are many unique aspects of ”C. burnetii” that made it an attractive target of weaponization research. Most notably, ”C. burnetii” maintains the lowest known pathogenic [[Minimal infective dose|infectious dose]], with some studies reporting that as low as one individual organism can incite infection in a human target.<ref>{{Citation |last1=Miller |first1=J. D. |title=Coxiella burnetii, Q Fever, and Bioterrorism |date=2006 |work=Microorganisms and Bioterrorism |pages=181–208 |editor-last=Anderson |editor-first=Burt |place=Boston, MA |publisher=Springer US |language=en |doi=10.1007/0-387-28159-2_10 |isbn=978-0-387-28159-9 |last2=Shaw |first2=E. I. |last3=Thompson |first3=H. A. |editor2-last=Friedman |editor2-first=Herman |editor3-last=Bendinelli |editor3-first=Mauro}}</ref> A World Health Organization (WHO) study estimated that releasing 50kg of ”C. burnetti” 2km upwind of a population of 500,000 human targets could result in around 150 deaths and incite symptomatic infections in more than 125,000 individuals.<ref>{{Cite web |last=CDC |date=2025-05-21 |title=Considerations for Intentional Release |url=https://www.cdc.gov/q-fever/hcp/intentionalrelease/index.html |access-date=2025-10-01 |website=Q Fever |language=en-us}}</ref> ”C. burnetii” has an [[incubation period]] of 1–3 weeks in the body between the point of infection and when symptoms can arise, posing a challenge for source tracing of the agent, enhancing its bioweapon potential. ”C. burnetii” can exist in two forms: a metabolically active large cell variant (LCV) and a spore-like small cell variant (SCV). The small cell variant is able to persist in the environment without a host while still maintaining infectious capabilities for years. Delicate enough to be dispersed miles by wind with negligible loss of virulence capabilities, the small cell variant is most responsible for dispersion of ”C. burnetii” across large areas and transmission of Q fever within populations.<ref>{{Cite journal |last1=McCaul |first1=T F |last2=Williams |first2=J C |date=September 1981 |title=Developmental cycle of Coxiella burnetii: structure and morphogenesis of vegetative and sporogenic differentiations |journal=Journal of Bacteriology |volume=147 |issue=3 |pages=1063–1076 |doi=10.1128/jb.147.3.1063-1076.1981 |pmid=7275931 |issn=0021-9193|pmc=216147 }}</ref>
The United States ended its [[biological warfare]] program in 1969. When it did, ”C. burnetii” was one of seven agents it had standardized as biological weapons.<ref name=croddyhart>{{cite book |last1=Croddy |first1=Eric C. |last2=Hart |first2=C. Perez-Armendariz J. |title=Chemical and Biological Warfare |url=https://books.google.com/books?id=fGp53P6ijqsC&pg=PA30 |publisher=Springer |year=2002 |pages=30–31 |isbn=0-387-95076-1 }}</ref> There are many unique aspects of ”C. burnetii” that made it an attractive target of weaponization research. Most notably, ”C. burnetii” maintains the lowest known pathogenic [[Minimal infective dose|infectious dose]], with some studies reporting that as low as one individual organism can incite infection in a human target.<ref>{{Citation |last1=Miller |first1=J. D. |title=Coxiella burnetii, Q Fever, and Bioterrorism |date=2006 |work=Microorganisms and Bioterrorism |pages=181–208 |editor-last=Anderson |editor-first=Burt |place=Boston, MA |publisher=Springer US |language=en |doi=10.1007/0-387-28159-2_10 |isbn=978-0-387-28159-9 |last2=Shaw |first2=E. I. |last3=Thompson |first3=H. A. |editor2-last=Friedman |editor2-first=Herman |editor3-last=Bendinelli |editor3-first=Mauro}}</ref> A World Health Organization (WHO) study estimated that releasing 50kg of ”C. burnetti” 2km upwind of a population of 500,000 human targets could result in around 150 deaths and incite symptomatic infections in more than 125,000 individuals.<ref>{{Cite web |last=CDC |date=2025-05-21 |title=Considerations for Intentional Release |url=https://www.cdc.gov/q-fever/hcp/intentionalrelease/index.html |access-date=2025-10-01 |website=Q Fever |language=en-us}}</ref> ”C. burnetii” has an [[incubation period]] of 1–3 weeks in the body between the point of infection and when symptoms can arise, posing a challenge for source tracing of the agent, enhancing its bioweapon potential. ”C. burnetii” can exist in two forms: a metabolically active large cell variant (LCV) and a spore-like small cell variant (SCV). The small cell variant is able to persist in the environment without a host while still maintaining infectious capabilities for years. Delicate enough to be dispersed miles by wind with negligible loss of virulence capabilities, the small cell variant is most responsible for dispersion of ”C. burnetii” across large areas and transmission of Q fever within populations.<ref>{{Cite journal |last1=McCaul |first1=T F |last2=Williams |first2=J C |date=September 1981 |title=Developmental cycle of Coxiella burnetii: structure and morphogenesis of vegetative and sporogenic differentiations |journal=Journal of Bacteriology |volume=147 |issue=3 |pages=1063–1076 |doi=10.1128/jb.147.3.1063-1076.1981 |pmid=7275931 |issn=0021-9193|pmc=216147 }}</ref>
Common ”C. burnetii” infections manifest as low grade flu-like symptoms, headache, [[malaise]], and persistent [[myalgia]].<ref>{{cite journal |last1=Hu |first1=Xueyuan |last2=Yu |first2=Yonghui |last3=Feng |first3=Junxia |last4=Fu |first4=Mengjiao |last5=Dai |first5=Lupeng |last6=Lu |first6=Zhiyu |last7=Luo |first7=Wenbo |last8=Wang |first8=Jinglin |last9=Zhou |first9=Dongsheng |last10=Xiong |first10=Xiaolu |last11=Wen |first11=Bohai |last12=Zhao |first12=Baohua |last13=Jiao |first13=Jun |title=Pathologic changes and immune responses against ”Coxiella burnetii” in mice following infection via non-invasive intratracheal inoculation |journal=PLOS ONE |date=2019-12-05 |volume=14 |issue=12 |article-number=e0225671 |doi=10.1371/journal.pone.0225671 |doi-access=free |issn=1932-6203 |pmc=6894818 |pmid=31805090 |bibcode=2019PLoSO..1425671H}}</ref> While the [[mortality rate]] for ”C. burnetii” is only around 0.5-1.5%, the bacteria can persist in the body for years and develop into chronic Q fever with further intense complications. Around 20% of those infected with ”C. burnetii” will experience chronic fatigue and symptoms that can last years or indefinitely after initial exposure”.” <ref>{{Cite web |title=Q Fever: Causes, Symptoms, Diagnosis, Prevention & Treatment |url=https://my.clevelandclinic.org/health/diseases/17883-q-fever |archive-url=http://web.archive.org/web/20250826114359/https://my.clevelandclinic.org/health/diseases/17883-q-fever |archive-date=2025-08-26 |access-date=2025-10-02 |website=Cleveland Clinic |language=en}}</ref> ”C. burnetii” is profiled as an infectious agent because of its ability to manifest as Q fever. Due to its low mortality but high [[morbidity]] rate, the Centers for Disease Control (CDC) has listed ”C. burnetii” as a Category B infectious agent. Category B is defined as being an agent that is moderately easy to disseminate and maintains lower lethality but more intense societal disruption.<ref>{{Cite journal |last1=Moodie |first1=Claire E. |last2=Thompson |first2=Herbert A. |last3=Meltzer |first3=Martin I. |last4=Swerdlow |first4=David L. |date=October 2008 |title=Prophylaxis after Exposure to Coxiella burnetii |volume=14 |issue=10 |journal=Emerging Infectious Diseases |pages=1558–1566 |language=en-us |doi=10.3201/eid1410.080576|pmid=18826819 |pmc=2609859 |doi-access=free }}</ref>
Common ”C. burnetii” infections manifest as low grade flu-like symptoms, headache, [[malaise]], and persistent [[myalgia]].<ref>{{cite journal |last1=Hu |first1=Xueyuan |last2=Yu |first2=Yonghui |last3=Feng |first3=Junxia |last4=Fu |first4=Mengjiao |last5=Dai |first5=Lupeng |last6=Lu |first6=Zhiyu |last7=Luo |first7=Wenbo |last8=Wang |first8=Jinglin |last9=Zhou |first9=Dongsheng |last10=Xiong |first10=Xiaolu |last11=Wen |first11=Bohai |last12=Zhao |first12=Baohua |last13=Jiao |first13=Jun |title=Pathologic changes and immune responses against ”Coxiella burnetii” in mice following infection via non-invasive intratracheal inoculation |journal=PLOS |date=2019-12-05 |volume=14 |issue=12 |article-number=e0225671 |doi=10.1371/journal.pone.0225671 |doi-access=free |issn=1932-6203 |pmc=6894818 |pmid=31805090 |bibcode=2019PLoSO..1425671H}}</ref> While the [[mortality rate]] for ”C. burnetii” is only around 0.5-1.5%, the bacteria can persist in the body for years and develop into chronic Q fever with further intense complications. Around 20% of those infected with ”C. burnetii” will experience chronic fatigue and symptoms that can last years or indefinitely after initial exposure”.” <ref>{{Cite web |title=Q Fever: Causes, Symptoms, Diagnosis, Prevention & Treatment |url=https://my.clevelandclinic.org/health/diseases/17883-q-fever |archive-url=http://web.archive.org/web/20250826114359/https://my.clevelandclinic.org/health/diseases/17883-q-fever |archive-date=2025-08-26 |access-date=2025-10-02 |website=Cleveland Clinic |language=en}}</ref> ”C. burnetii” is profiled as an infectious agent because of its ability to manifest as Q fever. Due to its low mortality but high [[morbidity]] rate, the Centers for Disease Control (CDC) has listed ”C. burnetii” as a Category B infectious agent. Category B is defined as being an agent that is moderately easy to disseminate and maintains lower lethality but more intense societal disruption.<ref>{{Cite journal |last1=Moodie |first1=Claire E. |last2=Thompson |first2=Herbert A. |last3=Meltzer |first3=Martin I. |last4=Swerdlow |first4=David L. |date=October 2008 |title=Prophylaxis after Exposure to Coxiella burnetii |volume=14 |issue=10 |journal=Emerging Infectious Diseases |pages=1558–1566 |language=en-us |doi=10.3201/eid1410.080576|pmid=18826819 |pmc=2609859 |doi-access=free }}</ref>
The United States, Iraq, and The Soviet Union were among the countries heading research into the potential use of ”C. burnetii” as a deliberately dispersed infectious agent in the mid-1900s.<ref>{{Cite journal |last=Anderson |first=Peter D. |last2=Bokor |first2=Gyula |date=2012-10-01 |title=Bioterrorism: Pathogens as Weapons |url=https://doi.org/10.1177/0897190012456366 |journal=Journal of Pharmacy Practice |language=EN |volume=25 |issue=5 |pages=521–529 |doi=10.1177/0897190012456366 |issn=0897-1900}}</ref> In the United States, ”C. burnetii” was field tested as an aerosolized infection in both animal and rodent trials at the Dugway Proving Ground in Utah, USA. Field testing allowed researchers the opportunity to observe and draw conclusions regarding the bacteria’s infectious dose, symptoms of disease, and progress into potential treatments. Testing of both rodent and human subjects within the same environment allowed for confirmation that ”C. burnetii” can be transmitted from animals to humans.<ref name=”:16″ /> [[File:Coxiella_burnetii_01.JPG|”C. burnetii”, the causative agent of [[Q fever]]|right|thumb]]
The United States, Iraq, and The Soviet Union were among the countries heading research into the potential use of ”C. burnetii” as a deliberately dispersed infectious agent in the mid-1900s.<ref>{{Cite journal |last=Anderson |first=Peter D. |last2=Bokor |first2=Gyula |date=2012-10-01 |title=Bioterrorism: Pathogens as Weapons |url=https://doi.org/10.1177/0897190012456366 |journal=Journal of Pharmacy Practice |language=EN |volume=25 |issue=5 |pages=521–529 |doi=10.1177/0897190012456366 |issn=0897-1900}}</ref> In the United States, ”C. burnetii” was field tested as an aerosolized infection in both animal and rodent trials at the Dugway Proving Ground in Utah, USA. Field testing allowed researchers the opportunity to observe and draw conclusions regarding the bacteria’s infectious dose, symptoms of disease, and progress into potential treatments. Testing of both rodent and human subjects within the same environment allowed for confirmation that ”C. burnetii” can be transmitted from animals to humans.<ref name=”:16″ /> [[File:Coxiella_burnetii_01.JPG|”C. burnetii”, the causative agent of [[Q fever]]|right|thumb]]
Recent advances in sequencing techniques, particularly selective whole genome amplification (SWGA), allowed for the recovery of even more ”C. burnetii” genomes. These samples were found in clinical and environmental studies. In one study, researchers applied SWGA to environmental samples such as unpasteurized milk and goat vaginal swabs. The technique helped increase the amount of ”C. burnetii” DNA found in the samples by up to 147 times more than before. This improvement dramatically increased bacterial reads and genome coverage from less than 1% of sequence reads to as high as 74%, providing enough genetic information to compare different strains and understand how they are related. The ability to sequence ”C. burnetii” even when it is present in very small amounts helps expand what we know about its genetic diversity and supports better tracking of how the bacteria spread and evolve.<ref>{{Cite journal |last1=Cocking |first1=Jill Hager |last2=Deberg |first2=Michael |last3=Schupp |first3=Jim |last4=Sahl |first4=Jason |last5=Wiggins |first5=Kristin |last6=Porty |first6=Ariel |last7=Hornstra |first7=Heidie M. |last8=Hepp |first8=Crystal |last9=Jardine |first9=Claire |last10=Furstenau |first10=Tara N. |last11=Schulte-Hostedde |first11=Albrecht |last12=Fofanov |first12=Viacheslav Y. |last13=Pearson |first13=Talima |date=2020-03-01 |title=Selective whole genome amplification and sequencing of Coxiella burnetii directly from environmental samples |journal=Genomics |volume=112 |issue=2 |pages=1872–1878 |doi=10.1016/j.ygeno.2019.10.022 |issn=0888-7543 |pmc=7199880 |pmid=31678592}}</ref>
Recent advances in sequencing techniques, particularly selective whole genome amplification (SWGA), allowed for the recovery of even more ”C. burnetii” genomes. These samples were found in clinical and environmental studies. In one study, researchers applied SWGA to environmental samples such as unpasteurized milk and goat vaginal swabs. The technique helped increase the amount of ”C. burnetii” DNA found in the samples by up to 147 times more than before. This improvement dramatically increased bacterial reads and genome coverage from less than 1% of sequence reads to as high as 74%, providing enough genetic information to compare different strains and understand how they are related. The ability to sequence ”C. burnetii” even when it is present in very small amounts helps expand what we know about its genetic diversity and supports better tracking of how the bacteria spread and evolve.<ref>{{Cite journal |last1=Cocking |first1=Jill Hager |last2=Deberg |first2=Michael |last3=Schupp |first3=Jim |last4=Sahl |first4=Jason |last5=Wiggins |first5=Kristin |last6=Porty |first6=Ariel |last7=Hornstra |first7=Heidie M. |last8=Hepp |first8=Crystal |last9=Jardine |first9=Claire |last10=Furstenau |first10=Tara N. |last11=Schulte-Hostedde |first11=Albrecht |last12=Fofanov |first12=Viacheslav Y. |last13=Pearson |first13=Talima |date=2020-03-01 |title=Selective whole genome amplification and sequencing of Coxiella burnetii directly from environmental samples |journal=Genomics |volume=112 |issue=2 |pages=1872–1878 |doi=10.1016/j.ygeno.2019.10.022 |issn=0888-7543 |pmc=7199880 |pmid=31678592}}</ref>
In bacteria, molecules called [[Bacterial small RNA|small regulatory RNAs]] are activated during stress and virulence conditions. In ”Coxiella burnetii,” several of these small RNAs (named CbSRs 1, 11, 12, and 14) are encoded within [[intergenic region]] (IGR), or the DNA between genes. CbSRs 2, 3, 4 and 9 are located complimentary to identified [[Open reading frame|ORF]]s. The CbSRs are up-regulated during intracellular growth in host cells.<ref>{{cite journal | vauthors = Warrier I, Hicks LD, Battisti JM, Raghavan R, Minnick MF | title = Identification of novel small RNAs and characterization of the 6S RNA of Coxiella burnetii | journal = PLOS ONE | volume = 9 | issue = 6 | article-number = e100147 | date = 2014 | pmid = 24949863 | pmc = 4064990 | doi = 10.1371/journal.pone.0100147 | bibcode = 2014PLoSO…9j0147W | doi-access = free }}</ref>
In bacteria, molecules called [[Bacterial small RNA|small regulatory RNAs]] are activated during stress and virulence conditions. In ”Coxiella burnetii,” several of these small RNAs (named CbSRs 1, 11, 12, and 14) are encoded within [[intergenic region]] (IGR), or the DNA between genes. CbSRs 2, 3, 4 and 9 are located complimentary to identified [[Open reading frame|ORF]]s. The CbSRs are up-regulated during intracellular growth in host cells.<ref>{{cite journal | vauthors = Warrier I, Hicks LD, Battisti JM, Raghavan R, Minnick MF | title = Identification of novel small RNAs and characterization of the 6S RNA of Coxiella burnetii | journal = PLOS | volume = 9 | issue = 6 | article-number = e100147 | date = 2014 | pmid = 24949863 | pmc = 4064990 | doi = 10.1371/journal.pone.0100147 | bibcode = 2014PLoSO…9j0147W | doi-access = free }}</ref>
All ”C. burnetii” strains contain extra genetic material – either one of four large plasmids (QpH1, QpDG, QpRS, or QpDV) or a piece of DNA in the chromosome that originated from the QpRS plasmid. The QpH1 plasmid plays a key role in helping the bacteria survive inside host cells such as mouse macrophages<ref>{{cite journal |last1=Luo |first1=Shengdong |last2=Lu |first2=Shanshan |last3=Fan |first3=Huahao |last4=Chen |first4=Zeliang |last5=Sun |first5=Zhihui |last6=Hu |first6=Yan |last7=Li |first7=Ruisheng |last8=An |first8=Xiaoping |last9=Uversky |first9=Vladimir N. |last10=Tong |first10=Yigang |last11=Song |first11=Lihua |title=The ”Coxiella burnetii” QpH1 Plasmid Is a Virulence Factor for Colonizing Bone Marrow-Derived Murine Macrophages |journal=Journal of Bacteriology |date=8 April 2021 |volume=203 |issue=9 |doi=10.1128/jb.00588-20|pmid=33558394 |pmc=8092169 }}</ref> and [[Vero cell]]s, although it is not needed for growth in artificial (axenic) culture. QpH1 also contains a [[toxin-antitoxin system]],<ref name=p36385554>{{cite journal |last1=Wachter |first1=S |last2=Cockrell |first2=DC |last3=Miller |first3=HE |last4=Virtaneva |first4=K |last5=Kanakabandi |first5=K |last6=Darwitz |first6=B |last7=Heinzen |first7=RA |last8=Beare |first8=PA |title=The endogenous ”Coxiella burnetii” plasmid encodes a functional toxin–antitoxin system. |journal=Molecular Microbiology |date=December 2022 |volume=118 |issue=6 |pages=744–764 |doi=10.1111/mmi.15001 |pmid=36385554 |pmc=10098735}}</ref> which may help the plasmid remain stable in the bacteria. Among all of these plasmids, eight conserved genes produce proteins that the bacteria inject into the host cells via the secretion system.<ref name=p36385554/>
All ”C. burnetii” strains contain extra genetic material – either one of four large plasmids (QpH1, QpDG, QpRS, or QpDV) or a piece of DNA in the chromosome that originated from the QpRS plasmid. The QpH1 plasmid plays a key role in helping the bacteria survive inside host cells such as mouse macrophages<ref>{{cite journal |last1=Luo |first1=Shengdong |last2=Lu |first2=Shanshan |last3=Fan |first3=Huahao |last4=Chen |first4=Zeliang |last5=Sun |first5=Zhihui |last6=Hu |first6=Yan |last7=Li |first7=Ruisheng |last8=An |first8=Xiaoping |last9=Uversky |first9=Vladimir N. |last10=Tong |first10=Yigang |last11=Song |first11=Lihua |title=The ”Coxiella burnetii” QpH1 Plasmid Is a Virulence Factor for Colonizing Bone Marrow-Derived Murine Macrophages |journal=Journal of Bacteriology |date=8 April 2021 |volume=203 |issue=9 |doi=10.1128/jb.00588-20|pmid=33558394 |pmc=8092169 }}</ref> and [[Vero cell]]s, although it is not needed for growth in artificial (axenic) culture. QpH1 also contains a [[toxin-antitoxin system]],<ref name=p36385554>{{cite journal |last1=Wachter |first1=S |last2=Cockrell |first2=DC |last3=Miller |first3=HE |last4=Virtaneva |first4=K |last5=Kanakabandi |first5=K |last6=Darwitz |first6=B |last7=Heinzen |first7=RA |last8=Beare |first8=PA |title=The endogenous ”Coxiella burnetii” plasmid encodes a functional toxin–antitoxin system. |journal=Molecular Microbiology |date=December 2022 |volume=118 |issue=6 |pages=744–764 |doi=10.1111/mmi.15001 |pmid=36385554 |pmc=10098735}}</ref> which may help the plasmid remain stable in the bacteria. Among all of these plasmids, eight conserved genes produce proteins that the bacteria inject into the host cells via the secretion system.<ref name=p36385554/>
Species of bacterium
Coxiella burnetii is a Gram-negative, obligate intracellular bacterial pathogen (a type of bacterium that lives inside the cells of its host) and the causative agent of Q fever. Although it was historically grouped with Rickettsia because of similar cell morphology, genetic and physiological studies show that C. burnetii is distinct and belongs to the class Gammaproteobacteria, and it is currently the only confirmed species within the genus Coxiella.[1] The bacterium is a small coccobacillus with a unique ability to survive in extremely harsh environmental conditions. It forms a highly resistant small-cell variant (SCV) as part of a two-stage developmental cycle, which alternates with a metabolically active large-cell variant (LCV) that replicates inside host cells.[2] These adaptations allow C. burnetii to survive common disinfectants and to persist within host cells, including in harsh environments such as the acidic phagolysosome.[1]
Coxiella burnetii was discovered in the 1930s through separate studies conducted in Montana, United States, and Queensland, Australia. Definitive descriptions of the pathogen were published later in the decade by Edward Holbrook Derrick and Macfarlane Burnet in Australia, and by Herald Rea Cox and Gordon Davis at the Rocky Mountain Laboratory (RML) in the United States.[3]
The RML team initially proposed the name Rickettsia diaporica, derived from the Greek word meaning “passing through,” in reference to the organism’s ability to pass through bacterial filters.[4] Around the same time, Edward Derrick suggested the name Rickettsia burnetii to recognize Macfarlane Burnet’s contribution in identifying the agent as a rickettsial organism.[3] As later research demonstrated that the species differed significantly from other rickettsiae, it was reassigned to a new subgenus, Coxiella. In 1948, Cornelius B. Philip, another RML researcher, promoted it to full genus status as Coxiella burnetii, honoring both Cox and Burnet.[3]
C. burnetii is now placed within the order Legionellales, and one of its closest known human-pathogenic relatives is Legionella pneumophila, the causative agent of Legionnaires’ disease.[5]
The first effective Q fever vaccine was developed in the 1960s–1970s through a collaboration between American microbiologist Richard Ormsbee and Australian infectious diseases physician Barrie Marmion.[6]
Coxiella was historically difficult to study because it could not be grown outside a host. In 2009, researchers established an axenic (host-free) culture system that allowed C. burnetii to be grown in cell-free media, enabling more detailed molecular studies of its biology.[7]
Virulence and infectious dose
[edit]
Of the many C. burnetii isolates, several strains are now used most often in research because they are well-characterized and widely available. The Nine Mile phase I strain remains the standard reference strain for genetic and virulence studies.[8] in addition, the Guiana strain has also become an important focus of recent research because it is more virulent in animal models than in the Nine Mile strain.[9] In recent years, additional strains have been examined, broadening the diversity of isolates used in research.[8] However, virulence can vary across host species, and murine rodents often require higher bacterial doses to establish infection in laboratory experiments.[10]
The median infectious dose (ID₅₀), the number of organisms required to infect 50% of exposed individuals, is estimated to be as low as a single bacterium when inhaled.[11] This represents an extremely low infectious dose (approximately 1 to 10 organisms), making Coxiella burnetii one of the most infectious bacterial pathogens known.[12] The disease occurs in two stages: an acute phase characterized by fever, chills, and respiratory symptoms, and a chronic phase that develops more slowly and may involve endocarditis or hepatitis.[1]
Cellular entry and infection mechanism
[edit]
C. burnetii infections begin in the alveoli of the lungs.[13] Upon inhalation, the bacterium targets alveolar macrophages and enters through actin-dependent phagocytosis.[3] After initial binding, it may enter non-professional phagocytes via an active zipper mechanism.[3] C. burnetii exploits the αVβ3 integrin receptor to enter host cells via actin- and RAC1-dependent phagocytosis, a process believed to help the pathogen avoid triggering inflammatory responses.[4]
Intracellular survival and development
[edit]
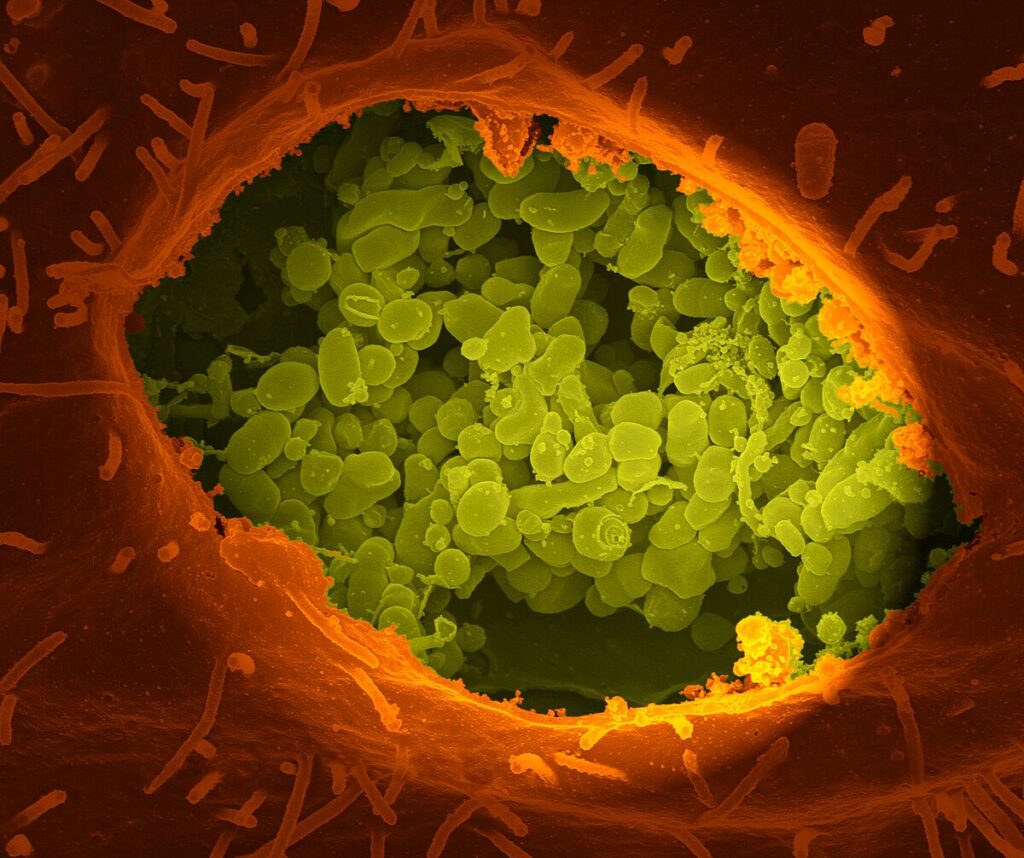
Following infection, C. burnetii undergoes a biphasic developmental cycle that alternates between two morphological forms: the small-cell variant (SCV) and the large-cell variant (LCV).[3] The SCV is metabolically dormant and highly resistant to environmental stressors, making it the form most likely responsible for initiating natural infection.[14]
After entry into a host cell, SCVs are taken up into a phagosome that matures through the endocytic pathway. In the first few hours after infection, the compartment merges with endosomes, autophagosomes, and lysosomes containing acid phosphatase picking up acidic hydrolases and lysosomal proteins along the way. The acidic environment inside the vacuole triggers the SVCs to transform into the metabolically active LCVs, which are then able to replicate within the C. burnetii-containing vacuole (CCV).[15]
At this stage, C. burnetii becomes metabolically active and synthesizes a type IV secretion system (T4SS) which translocates effector proteins into the host cytoplasm to manipulate host cell functions.[12] After approximately six days, the bacterium transitions back to the SCV form to prepare for persistence and extracellular release.[10][16]
Type IVB secretion system
[edit]
The bacteria use a type IVB secretion system known as Icm/Dot (intracellular multiplication / defect in organelle trafficking genes) to inject over 100 effector proteins into the host.[17][18] These effectors increase the bacterium’s ability to survive and replicate within host cells by modulating multiple host pathways, including blocking apoptosis, inhibiting immune responses, and altering vesicle trafficking.[16][19] In Legionella pneumophila, a related Gammaproteobacterium that uses the same secretion system, these effectors enhance survival by preventing fusion of the bacteria-containing vacuole with degradative endosomes.[20]
Treatment and prevention
[edit]
Acute Q fever is typically treated with doxycycline, which shortens illness duration and reduces the risk of progression to chronic disease. Other antibiotics, including macrolides, co-trimoxazole, quinolones, and beta-lactams, have been used but show less consistent effectiveness. Serologic tests may not become positive until 1–2 weeks after exposure, so the CDC recommends initiating doxycycline based on clinical suspicion rather than waiting for laboratory confirmation.[21][22]
Chronic Q fever, particularly endocarditis, requires prolonged combination therapy. The most effective regimen is doxycycline with hydroxychloroquine for at least 18 months, and patients undergoing treatment require long-term serologic monitoring to ensure clearance of infection and detect relapse.[21]
Despite ongoing research into new human Q fever vaccines, Q-VAX® remains the only commercially available vaccine and is not licensed for use in the United States.[23] Prevention mainly involves avoiding exposure to infected animals and raw milk products. Individuals with certain underlying conditions may also require closer monitoring due to a higher risk of chronic infection.[24]
Prevalence and host
[edit]
Coxiella burnetii is a globally distributed microbe (excluding New Zealand and Antarctica) found commonly in domestic reservoirs such as sheep, goats, and cattle.[25] The microbe is spread to other organisms through the inhalation of contaminated aerosols. It presents asymptomatically in animals, and proves to cause chronic disease in nearby humans. The microbe is spread from domestic animals to human hosts during parturition or slaughter of the domestic animal.[26] Ticks can spread the microbe across generations of livestock through transovarial transmission. Other insects such as flies, cockroaches, and lice can also act as reservoirs.[25]
Q fever is present in German human populations with 27-100 cases reported annually. It is estimated to affect 50 per every 100,000 inhabitants annually in France. The UK has faced 904 cases between 2000 and 2015, as well as major outbreaks in 2002 and 2007. Italy has experienced two different outbreaks of human infection in Como prison and Vicenza respectively. Spain has a high prevalence of C. burnetii infection in human populations in Basque and Navarre. The annual reported cases of human C. burnetii infection in the US went up from 19 cases in 2000 to around 160-180 cases per year, with 178 acute cases in 2019. In the US, cases of human infection were mainly found in the West or the Great Plains regions of the country. Canada has a low human incidence of infection. In Quebec, it was reported that there were 0.4 cases per 100,000 inhabitants in 2017. In Alberta, only 39 cases were reported.[26] Coxiella burnetii has been observed with a greater prevalence in populations in close proximity to animals as well as homeless populations in Brazil.[27] In Algeria, residents of rural areas were at a three times higher risk for Coxiella burnetii infections compared to inhabitants of urban areas.[28] Isiolo County, Kenya has been found to have a 44.7% seroprevalence of Coxiella burnetii in humans.[29] In Kwara State, Nigeria, C. burnetii was found with a 18.8% prevalence in milk and cheese, contributing to further transmission of Q fever through consumption of milk products.[30]
Coxiella burnetii and bacteria similar to it were found in tick species that populate wildlife in South Korea.[31] The microbe has also been found in ticks that were collected from Churra Galega Mirandesa sheep in Portugal.[32] Isiolo County, Kenya has been found to have a 47.9% seroprevalence of C. burnetii in livestock.[29] The microbe has also been found in populations of water buffalo on the Nile Delta in Egypt. The highest seroprevalence found in Egypt was 15.5% in the city of Kafr El-Sheikh.[25]
Historical assessment of bioweapon potential
[edit]
The United States ended its biological warfare program in 1969. When it did, C. burnetii was one of seven agents it had standardized as biological weapons.[33] There are many unique aspects of C. burnetii that made it an attractive target of weaponization research. Most notably, C. burnetii maintains the lowest known pathogenic infectious dose, with some studies reporting that as low as one individual organism can incite infection in a human target.[34] A World Health Organization (WHO) study estimated that releasing 50kg of C. burnetti 2km upwind of a population of 500,000 human targets could result in around 150 deaths and incite symptomatic infections in more than 125,000 individuals.[35] C. burnetii has an incubation period of 1–3 weeks in the body between the point of infection and when symptoms can arise, posing a challenge for source tracing of the agent, enhancing its bioweapon potential. C. burnetii can exist in two forms: a metabolically active large cell variant (LCV) and a spore-like small cell variant (SCV). The small cell variant is able to persist in the environment without a host while still maintaining infectious capabilities for years. Delicate enough to be dispersed miles by wind with negligible loss of virulence capabilities, the small cell variant is most responsible for dispersion of C. burnetii across large areas and transmission of Q fever within populations.[36]
Common C. burnetii infections manifest as low grade flu-like symptoms, headache, malaise, and persistent myalgia.[37] While the mortality rate for C. burnetii is only around 0.5-1.5%, the bacteria can persist in the body for years and develop into chronic Q fever with further intense complications. Around 20% of those infected with C. burnetii will experience chronic fatigue and symptoms that can last years or indefinitely after initial exposure. [38] C. burnetii is profiled as an infectious agent because of its ability to manifest as Q fever. Due to its low mortality but high morbidity rate, the Centers for Disease Control (CDC) has listed C. burnetii as a Category B infectious agent. Category B is defined as being an agent that is moderately easy to disseminate and maintains lower lethality but more intense societal disruption.[39]
The United States, Iraq, and The Soviet Union were among the countries heading research into the potential use of C. burnetii as a deliberately dispersed infectious agent in the mid-1900s.[40] In the United States, C. burnetii was field tested as an aerosolized infection in both animal and rodent trials at the Dugway Proving Ground in Utah, USA. Field testing allowed researchers the opportunity to observe and draw conclusions regarding the bacteria’s infectious dose, symptoms of disease, and progress into potential treatments. Testing of both rodent and human subjects within the same environment allowed for confirmation that C. burnetii can be transmitted from animals to humans.[16]
At least 75 completely sequenced genomes of Coxiella burnetii strains exist,[41] which contain about 2.1 million base pairs of DNA each and encode around 2,100 open reading frames (ORFs), which are stretches of DNA that can be read to produce proteins; 746 (or about 35%) of these genes have no known function.
Recent advances in sequencing techniques, particularly selective whole genome amplification (SWGA), allowed for the recovery of even more C. burnetii genomes. These samples were found in clinical and environmental studies. In one study, researchers applied SWGA to environmental samples such as unpasteurized milk and goat vaginal swabs. The technique helped increase the amount of C. burnetii DNA found in the samples by up to 147 times more than before. This improvement dramatically increased bacterial reads and genome coverage from less than 1% of sequence reads to as high as 74%, providing enough genetic information to compare different strains and understand how they are related. The ability to sequence C. burnetii even when it is present in very small amounts helps expand what we know about its genetic diversity and supports better tracking of how the bacteria spread and evolve.[42]
In bacteria, molecules called small regulatory RNAs are activated during stress and virulence conditions. In Coxiella burnetii, several of these small RNAs (named CbSRs 1, 11, 12, and 14) are encoded within intergenic region (IGR), or the DNA between genes. CbSRs 2, 3, 4 and 9 are located complimentary to identified ORFs. The CbSRs are up-regulated during intracellular growth in host cells.[43]
All C. burnetii strains contain extra genetic material – either one of four large plasmids (QpH1, QpDG, QpRS, or QpDV) or a piece of DNA in the chromosome that originated from the QpRS plasmid. The QpH1 plasmid plays a key role in helping the bacteria survive inside host cells such as mouse macrophages[44] and Vero cells, although it is not needed for growth in artificial (axenic) culture. QpH1 also contains a toxin-antitoxin system,[45] which may help the plasmid remain stable in the bacteria. Among all of these plasmids, eight conserved genes produce proteins that the bacteria inject into the host cells via the secretion system.[45]
One Health implications and significance
[edit]
Coxiella burnetii is recognized as one of the most widely distributed zoonotic pathogens in the world and the causative agent of human Q fever. One Health is an approach that emphasizes the interconnectedness of environmental, animal, and human health.[46] Due to its zoonotic transmission pathways and exceptional environmental persistence, C. burnetii is regarded as a priority pathogen within the One Health approach.[47] Its ability to resist environmental stressors and infect a broad range of hosts allows the circulation of C. burnetii between diverse hosts and environments.[16] C. burnetii can be transmitted from animals to humans, with livestock serving as the primary reservoir responsible for most human infections. Livestock, mainly sheep, goats, and cattle can remain relatively asymptomatic upon infection, facilitating undetected transmission of C. burnetii between hosts.[48] Rural populations are often in closer contact with C. burnetii than urban or suburban communities due to proximity to livestock.[49] A range of antibiotics and combination therapies have been found to be effective at clearing individual infections after detection and onset of symptoms. The Q-Vax vaccine can provide effective prevention of an initial infection, although this vaccine has not been widely implemented due to high incidence of a post-vaccination hypersensitivity reaction. Ongoing research is focussed on remodelling this vaccine to ensure that side effects are minimized and widespread deployment is successful.[50] The global distribution of C. burnetii can be attributed to its uniquely resilient small cell variant that is able to be dispersed miles by wind and persist in its environment for years. Diagnosis of a C. burnetii infection remains difficult, as its acute manifestation can resemble that of many other non-specific and self-limiting infectious diseases.[51] Control of outbreaks is further complicated by the pathogens environmental resilience, capacity for aerosol transmission, and widespread distribution. Lack of a widely approved vaccine and the difficulty of detecting C. burnetii make it an ongoing subject of public health and infectious disease research.[52]
- ^ a b c Shaw, Edward I.; Voth, Daniel E. (January 2019). “Coxiella burnetii: A Pathogenic Intracellular Acidophile”. Microbiology (Reading, England). 165 (1): 1–3. doi:10.1099/mic.0.000707. ISSN 1465-2080. PMC 6600347. PMID 30422108.
- ^ Shepherd, Doulin C.; Kaplan, Mohammed; Vankadari, Naveen; Kim, Ki Woo; Larson, Charles L.; Dutka, Przemysław; Beare, Paul A.; Krzymowski, Edward; Heinzen, Robert A.; Jensen, Grant J.; Ghosal, Debnath (2023-07-21). “Morphological remodeling of Coxiella burnetii during its biphasic developmental cycle revealed by cryo-electron tomography”. iScience. 26 (7) 107210. Bibcode:2023iSci…26j7210S. doi:10.1016/j.isci.2023.107210. ISSN 2589-0042. PMC 10362272. PMID 37485371.
- ^ a b c d e f Roest, Hendrik I.J.; Bossers, Alex; van Zijderveld, Fred G.; Rebel, Johanna M.L. (2013-09-01). “Clinical microbiology of Coxiella burnetii and relevant aspects for the diagnosis and control of the zoonotic disease Q fever”. Veterinary Quarterly. 33 (3): 148–160. doi:10.1080/01652176.2013.843809. ISSN 0165-2176. PMID 24161079.
- ^ a b Hirschmann, J. V. (2019-07-01). “The Discovery of Q Fever and Its Cause”. The American Journal of the Medical Sciences. 358 (1): 3–10. doi:10.1016/j.amjms.2019.04.006. ISSN 0002-9629. PMID 31076071.
- ^ Duron, Olivier; Doublet, Patricia; Vavre, Fabrice; Bouchon, Didier (2018-12-01). “The Importance of Revisiting Legionellales Diversity”. Trends in Parasitology. 34 (12): 1027–1037. doi:10.1016/j.pt.2018.09.008. ISSN 1471-4922. PMID 30322750.
- ^ Graves, Stephen R. (2025-10-30). “The Story behind the Science: A history of Q-VAX, the Australian human vaccine against Q fever”. mBio. 0 e00419-25: e00419–25. doi:10.1128/mbio.00419-25. PMID 41165325.
- ^ Vallejo Esquerra, Eduardo; Yang, Hong; Sanchez, Savannah E.; Omsland, Anders (2017-05-31). “Physicochemical and Nutritional Requirements for Axenic Replication Suggest Physiological Basis for Coxiella burnetii Niche Restriction”. Frontiers in Cellular and Infection Microbiology. 7 190. doi:10.3389/fcimb.2017.00190. ISSN 2235-2988. PMC 5449765. PMID 28620582.
- ^ a b Abou Abdallah, Rita; Million, Matthieu; Delerce, Jeremy; Anani, Hussein; Diop, Awa; Caputo, Aurelia; Zgheib, Rita; Rousset, Elodie; Sidi Boumedine, Karim; Raoult, Didier; Fournier, Pierre-Edouard (2022-11-21). “Pangenomic analysis of Coxiella burnetii unveils new traits in genome architecture”. Frontiers in Microbiology. 13 1022356. doi:10.3389/fmicb.2022.1022356. ISSN 1664-302X. PMID 36478861.
- ^ Melenotte, C.; Caputo, A.; Bechah, Y.; Lepidi, H.; Terras, J.; Kowalczewska, M.; Di Pinto, F.; Nappez, C.; Raoult, D.; Brégeon, F. (September 2019). “The hypervirulent Coxiella burnetii Guiana strain compared in silico, in vitro and in vivo to the Nine Mile and the German strain”. Clinical Microbiology and Infection: The Official Publication of the European Society of Clinical Microbiology and Infectious Diseases. 25 (9): 1155.e1–1155.e8. doi:10.1016/j.cmi.2018.12.039. ISSN 1469-0691. PMID 30625413.
- ^ a b Eldin, Carole; Mélenotte, Cléa; Mediannikov, Oleg; Ghigo, Eric; Million, Matthieu; Edouard, Sophie; Mege, Jean-Louis; Maurin, Max; Raoult, Didier (January 2017). “From Q Fever to Coxiella burnetii Infection: a Paradigm Change”. Clinical Microbiology Reviews. 30 (1): 115–190. doi:10.1128/CMR.00045-16. ISSN 0893-8512. PMC 5217791. PMID 27856520.
- ^ Asghar, Faiza; Hayek, Inaya; Berens, Christian; Liebler-Tenorio, Elisabeth; Lührmann, Anja (2025-10-13), Coxiella burnetii establishes a small cell variant (SCV)-like persistent form to survive adverse intracellular conditions, bioRxiv, doi:10.1101/2025.09.18.677042, retrieved 2025-11-14
- ^ a b “Q fever caused by Coxiella burnetii”. Centers for Disease Control. 15 January 2019.
- ^ Polo, M. F.; Mastrandrea, S.; Santoru, L.; Arcadu, A.; Masala, G.; Marras, V.; Bagella, G.; Sechi, M. M.; Tanda, F.; Pirina, P. (2015-11-01). “Pulmonary inflammatory pseudotumor due to Coxiella burnetii. Case report and literature review”. Microbes and Infection. Special issue on intracellular bacteria. 17 (11): 795–798. doi:10.1016/j.micinf.2015.08.008. ISSN 1286-4579.
- ^ Saxon, Wolfgang (March 8, 2001). “Dr. Paul Fiset, 78, Microbiologist And Developer of Q Fever Vaccine”. New York Times. p. C-17.
- ^ Clemente, Tatiana M.; Angara, Rajendra K.; Gilk, Stacey D. (2023-08-14). “Establishing the intracellular niche of obligate intracellular vacuolar pathogens”. Frontiers in Cellular and Infection Microbiology. 13 1206037. doi:10.3389/fcimb.2023.1206037. ISSN 2235-2988. PMC 10461009. PMID 37645379.
- ^ a b c d Dragan, Amanda L.; Voth, Daniel E. (April 2020). “Coxiella burnetii: international pathogen of mystery”. Microbes and Infection. 22 (3): 100–110. doi:10.1016/j.micinf.2019.09.001. ISSN 1286-4579. PMC 7101257. PMID 31574310.
- ^ van Schaik, Erin J.; Chen, Chen; Mertens, Katja; Weber, Mary M.; Samuel, James E. (August 2013). “Molecular pathogenesis of the obligate intracellular bacterium Coxiella burnetii”. Nature Reviews. Microbiology. 11 (8): 561–573. doi:10.1038/nrmicro3049. ISSN 1740-1534. PMC 4134018. PMID 23797173.
- ^ Newton, Hayley J.; Kohler, Lara J.; McDonough, Justin A.; Temoche-Diaz, Morayma; Crabill, Emerson; Hartland, Elizabeth L.; Roy, Craig R. (July 2014). “A screen of Coxiella burnetii mutants reveals important roles for Dot/Icm effectors and host autophagy in vacuole biogenesis”. PLOS Pathogens. 10 (7) e1004286. doi:10.1371/journal.ppat.1004286. ISSN 1553-7374. PMC 4117601. PMID 25080348.
- ^ Lührmann, Anja; Nogueira, Catarina V.; Carey, Kimberly L.; Roy, Craig R. (2010-11-02). “Inhibition of pathogen-induced apoptosis by a Coxiella burnetii type IV effector protein”. Proceedings of the National Academy of Sciences of the United States of America. 107 (44): 18997–19001. Bibcode:2010PNAS..10718997L. doi:10.1073/pnas.1004380107. ISSN 1091-6490. PMC 2973885. PMID 20944063.
- ^ Pan, Xiaoxiao; Lührmann, Anja; Satoh, Ayano; Laskowski-Arce, Michelle A.; Roy, Craig R. (2008-06-20). “Ankyrin repeat proteins comprise a diverse family of bacterial type IV effectors”. Science (New York, N.Y.). 320 (5883): 1651–1654. Bibcode:2008Sci…320.1651P. doi:10.1126/science.1158160. ISSN 1095-9203. PMC 2514061. PMID 18566289.
- ^ a b Kersh, Gilbert J. (November 2013). “Antimicrobial therapies for Q fever”. Expert Review of Anti-Infective Therapy. 11 (11): 1207–1214. doi:10.1586/14787210.2013.840534. ISSN 1744-8336. PMC 4608426. PMID 24073941.
- ^ CDC (2025-05-21). “Clinical Guidance for Q fever”. Q Fever. Retrieved 2025-11-19.
- ^ Long, Carrie Mae (2021-09-22). “Q Fever Vaccine Development: Current Strategies and Future Considerations”. Pathogens (Basel, Switzerland). 10 (10): 1223. doi:10.3390/pathogens10101223. ISSN 2076-0817. PMC 8539696. PMID 34684172.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ CDC (2025-05-21). “About Q fever”. Q Fever. Retrieved 2025-11-19.
- ^ a b c Selim, Abdelfattah; Marzok, Mohamed; Gattan, Hattan S.; Ismail, Hesham (2025-10-07). “Risk factors influencing Coxiella burnetii seropositivity in water Buffalo (Bubalus bubalis) populations of Egypt’s Nile Delta”. PLOS One. 20 (10) e0333680. Bibcode:2025PLoSO..2033680S. doi:10.1371/journal.pone.0333680. ISSN 1932-6203. PMC 12503306. PMID 41056260.
- ^ a b Rahal, Mohamed; Salhi, Omar; Ouchetati, Imane; Khelifi Touhami, Nadjet Amina; Ouchene, Nassim (2025-11-01). “Global epidemiology and molecular typing of Coxiella burnetii: A systematic review of Q fever in humans and animals”. Comparative Immunology, Microbiology and Infectious Diseases. 123 102401. doi:10.1016/j.cimid.2025.102401. ISSN 0147-9571. PMID 40902447.
- ^ de França, Danilo Alves; Kmetiuk, Louise Bach; do Couto, Anahi Chechia; Langoni, Helio; Biondo, Alexander Welker (2025-08-03). “Coxiella burnetii and HIV infection in people experiencing homelessness”. Scientific Reports. 15 (1) 28312. Bibcode:2025NatSR..1528312D. doi:10.1038/s41598-025-09422-z. ISSN 2045-2322. PMC 12319078. PMID 40754629.
- ^ Lacheheb, A.; Raoult, D. (2009-12-01). “Seroprevalence of Q-fever in Algeria”. Clinical Microbiology and Infection. 15: 167–168. doi:10.1111/j.1469-0691.2008.02211.x. ISSN 1198-743X. PMID 19793121.
- ^ a b Mutisya, Wilfred Mutuku; Akoko, James M.; Mwatondo, Athman; Muturi, Mathew; Nthiwa, Daniel; Abkallo, Hussein M.; Nyamota, Richard; Wachira, Timothy; Gathura, Peter; Bett, Bernard (2025-10-17). “Sero-epidemiology of Coxiella burnetii in livestock and humans in Isiolo county Kenya”. PLOS Neglected Tropical Diseases. 19 (10) e0013557. doi:10.1371/journal.pntd.0013557. ISSN 1935-2735. PMC 12551958. PMID 41105740.
- ^ Elelu, Nusirat; Chinedu, Nwachukwu Raymond; Yakub-Obalowu, Balkees A.; Odetokun, Ismail A.; Al-Mustapha, Ahmad I. (2025-01-07). “Real-time PCR detection of Coxiella burnetii in dairy products in Kwara State, Nigeria: a public health concern”. BMC Veterinary Research. 21 (1): 8. doi:10.1186/s12917-024-04449-2. ISSN 1746-6148. PMC 11706007. PMID 39773743.
- ^ Lee, You-Jeong; Chae, Su-Jin; Kim, Beoul; Shin, Hak Sub; Kwak, Sun Min; Jeong, Hyesung; Lee, Suwoong; Kang, Yong-Myung; Kwak, Dongmi; Seo, Min-Goo (January 2025). Ferrara, Gianmarco (ed.). “First Molecular Typing of Tick-Derived Coxiella burnetii From Wildlife in South Korea: Surveillance and Genetic Characterization”. Transboundary and Emerging Diseases. 2025 (1) 2533438. doi:10.1155/tbed/2533438. ISSN 1865-1674. PMC 12513786. PMID 41080909.
- ^ Gomes-Gonçalves, Sara; Mesquita, João R.; Ruano, Zita Martins; Barradas, Patrícia F. (2025-08-01). “Coxiella burnetii detected in Rhipicephalus sanguineus ticks collected in Portuguese autochthonous livestock”. Acta Tropica. 268 107697. doi:10.1016/j.actatropica.2025.107697. ISSN 0001-706X. PMID 40523586.
- ^ Croddy, Eric C.; Hart, C. Perez-Armendariz J. (2002). Chemical and Biological Warfare. Springer. pp. 30–31. ISBN 0-387-95076-1.
- ^ Miller, J. D.; Shaw, E. I.; Thompson, H. A. (2006), Anderson, Burt; Friedman, Herman; Bendinelli, Mauro (eds.), “Coxiella burnetii, Q Fever, and Bioterrorism”, Microorganisms and Bioterrorism, Boston, MA: Springer US, pp. 181–208, doi:10.1007/0-387-28159-2_10, ISBN 978-0-387-28159-9
- ^ CDC (2025-05-21). “Considerations for Intentional Release”. Q Fever. Retrieved 2025-10-01.
- ^ McCaul, T F; Williams, J C (September 1981). “Developmental cycle of Coxiella burnetii: structure and morphogenesis of vegetative and sporogenic differentiations”. Journal of Bacteriology. 147 (3): 1063–1076. doi:10.1128/jb.147.3.1063-1076.1981. ISSN 0021-9193. PMC 216147. PMID 7275931.
- ^ Hu, Xueyuan; Yu, Yonghui; Feng, Junxia; Fu, Mengjiao; Dai, Lupeng; Lu, Zhiyu; Luo, Wenbo; Wang, Jinglin; Zhou, Dongsheng; Xiong, Xiaolu; Wen, Bohai; Zhao, Baohua; Jiao, Jun (2019-12-05). “Pathologic changes and immune responses against Coxiella burnetii in mice following infection via non-invasive intratracheal inoculation”. PLOS One. 14 (12) e0225671. Bibcode:2019PLoSO..1425671H. doi:10.1371/journal.pone.0225671. ISSN 1932-6203. PMC 6894818. PMID 31805090.
- ^ “Q Fever: Causes, Symptoms, Diagnosis, Prevention & Treatment”. Cleveland Clinic. Archived from the original on 2025-08-26. Retrieved 2025-10-02.
- ^ Moodie, Claire E.; Thompson, Herbert A.; Meltzer, Martin I.; Swerdlow, David L. (October 2008). “Prophylaxis after Exposure to Coxiella burnetii”. Emerging Infectious Diseases. 14 (10): 1558–1566. doi:10.3201/eid1410.080576. PMC 2609859. PMID 18826819.
- ^ Anderson, Peter D.; Bokor, Gyula (2012-10-01). “Bioterrorism: Pathogens as Weapons”. Journal of Pharmacy Practice. 25 (5): 521–529. doi:10.1177/0897190012456366. ISSN 0897-1900.
- ^ Abou Abdallah, Rita; Million, Matthieu; Delerce, Jeremy; Anani, Hussein; Diop, Awa; Caputo, Aurelia; Zgheib, Rita; Rousset, Elodie; Sidi Boumedine, Karim; Raoult, Didier; Fournier, Pierre-Edouard (2022). “Pangenomic analysis of Coxiella burnetii unveils new traits in genome architecture”. Frontiers in Microbiology. 13 1022356. doi:10.3389/fmicb.2022.1022356. ISSN 1664-302X. PMC 9721466. PMID 36478861.
- ^ Cocking, Jill Hager; Deberg, Michael; Schupp, Jim; Sahl, Jason; Wiggins, Kristin; Porty, Ariel; Hornstra, Heidie M.; Hepp, Crystal; Jardine, Claire; Furstenau, Tara N.; Schulte-Hostedde, Albrecht; Fofanov, Viacheslav Y.; Pearson, Talima (2020-03-01). “Selective whole genome amplification and sequencing of Coxiella burnetii directly from environmental samples”. Genomics. 112 (2): 1872–1878. doi:10.1016/j.ygeno.2019.10.022. ISSN 0888-7543. PMC 7199880. PMID 31678592.
- ^ Warrier I, Hicks LD, Battisti JM, Raghavan R, Minnick MF (2014). “Identification of novel small RNAs and characterization of the 6S RNA of Coxiella burnetii”. PLOS One. 9 (6) e100147. Bibcode:2014PLoSO…9j0147W. doi:10.1371/journal.pone.0100147. PMC 4064990. PMID 24949863.
- ^ Luo, Shengdong; Lu, Shanshan; Fan, Huahao; Chen, Zeliang; Sun, Zhihui; Hu, Yan; Li, Ruisheng; An, Xiaoping; Uversky, Vladimir N.; Tong, Yigang; Song, Lihua (8 April 2021). “The Coxiella burnetii QpH1 Plasmid Is a Virulence Factor for Colonizing Bone Marrow-Derived Murine Macrophages”. Journal of Bacteriology. 203 (9). doi:10.1128/jb.00588-20. PMC 8092169. PMID 33558394.
- ^ a b Wachter, S; Cockrell, DC; Miller, HE; Virtaneva, K; Kanakabandi, K; Darwitz, B; Heinzen, RA; Beare, PA (December 2022). “The endogenous Coxiella burnetii plasmid encodes a functional toxin–antitoxin system”. Molecular Microbiology. 118 (6): 744–764. doi:10.1111/mmi.15001. PMC 10098735. PMID 36385554.
- ^ Lerner, Henrik; Berg, Charlotte (2015-01-01). “The concept of health in One Health and some practical implications for research and education: what is One Health?”. Infection Ecology & Epidemiology. 5 (1) 25300. Bibcode:2015InfEE…525300L. doi:10.3402/iee.v5.25300. PMC 4320999. PMID 25660757.
- ^ Bauer, Benjamin U.; Knittler, Michael R.; Andrack, Jennifer; Berens, Christian; Campe, Amely; Christiansen, Bahne; Fasemore, Akinyemi M.; Fischer, Silke F.; Ganter, Martin; Körner, Sophia; Makert, Gustavo R.; Matthiesen, Svea; Mertens-Scholz, Katja; Rinkel, Sven; Runge, Martin (2023-11-01). “Interdisciplinary studies on Coxiella burnetii: From molecular to cellular, to host, to one health research”. International Journal of Medical Microbiology. 313 (6) 151590. doi:10.1016/j.ijmm.2023.151590. ISSN 1438-4221. PMID 38056089.
- ^ Muhammad, Kaka A.; Gadzama, Usman N.; Onyiche, ThankGod E. (2023-10-01). “Distribution and Prevalence of Coxiella burnetii in Animals, Humans, and Ticks in Nigeria: A Systematic Review”. Infectious Disease Reports. 15 (5): 576–588. doi:10.3390/idr15050056. ISSN 2036-7430. PMC 10606657. PMID 37888137.
- ^ González-Barrio, David; Ruiz-Fons, Francisco (2019). “Coxiella burnetii in wild mammals: A systematic review”. Transboundary and Emerging Diseases. 66 (2): 662–671. doi:10.1111/tbed.13085. ISSN 1865-1682. PMID 30506629.
- ^ Heinzen, Robert A.; Pierce, Kathleen N.; Voth, Daniel E.; Stenos, John; Graves, Stephen; Long, Carrie Mae (2025-11-06). Jensen, Kirk D. C. (ed.). “Control of human Q fever by vaccination: the journey to Q-VAX and beyond”. Infection and Immunity e00280-25. doi:10.1128/iai.00280-25. ISSN 0019-9567. PMID 41196302.
- ^ Kazar, Jan (2005). “Coxiella burnetii Infection”. Annals of the New York Academy of Sciences. 1063 (1): 105–114. Bibcode:2005NYASA1063..105K. doi:10.1196/annals.1355.018. ISSN 1749-6632. PMID 16481501.
- ^ Mohammadi, Mohammad Reza; Moradkasani, Safoura; Latifian, Mina; Esmaeili, Saber (2025-10-01). “Coxiella burnetii: Emerging threats, molecular insights, and advances in diagnosis and control measures”. Journal of Microbiological Methods. 237 107213. doi:10.1016/j.mimet.2025.107213. ISSN 0167-7012.