From Wikipedia, the free encyclopedia
Content deleted Content added
| Line 73: | Line 73: | ||
|
[[Category:Cardiac myosin inhibitors]] |
[[Category:Cardiac myosin inhibitors]] |
||
|
[[Category:Pyridopyrimidines]] |
[[Category:Pyridopyrimidines]] |
||
|
[[Category:2-Fluorophenyl compounds]] |
|||
Revision as of 07:01, 18 October 2025
Cardiac myosin inhibitor developed by Bristol Myers Squibb
Pharmaceutical compound
 |
|
| Other names | MYK-224; BMS-986435 |
|---|---|
|
|
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
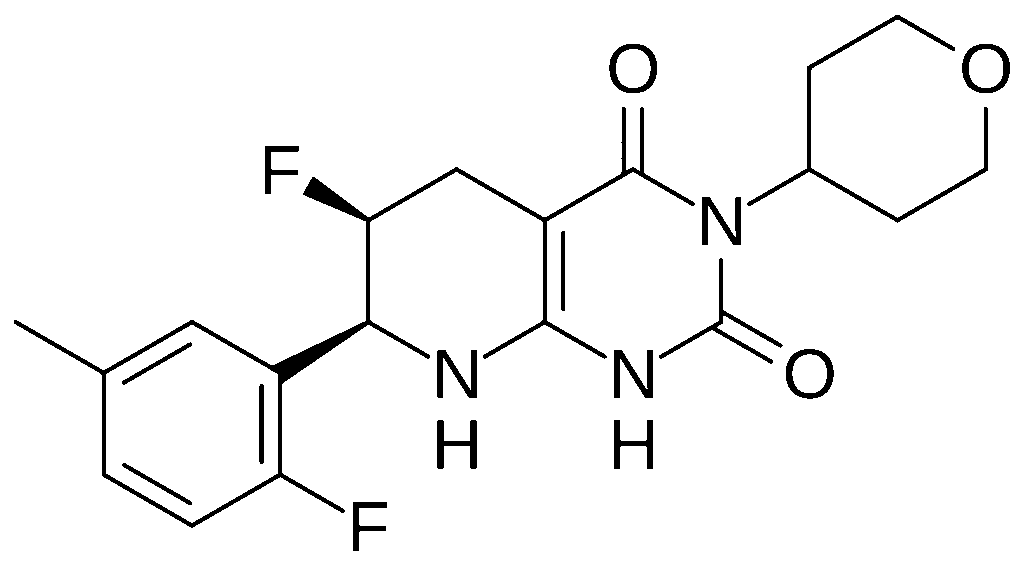
| Formula | C19H21F2N3O3 |
| Molar mass | 377.392 g·mol−1 |
| 3D model (JSmol) | |
|
|
|
|
Delocamten (development code MYK-224) is a small-molecule cardiac myosin inhibitor developed by Bristol Myers Squibb for hypertrophic cardiomyopathy.[1][2][3]
References
- ^ Lehman, Sarah J.; Crocini, Claudia; Leinwand, Leslie A. (June 2022). “Targeting the sarcomere in inherited cardiomyopathies”. Nature Reviews Cardiology. 19 (6): 353–363. doi:10.1038/s41569-022-00682-0. ISSN 1759-5010. PMC 9119933. PMID 35304599.
- ^ Sebastian, Sneha Annie; Padda, Inderbir; Lehr, Eric J.; Johal, Gurpreet (September 2023). “Aficamten: A Breakthrough Therapy for Symptomatic Obstructive Hypertrophic Cardiomyopathy”. American Journal of Cardiovascular Drugs. 23 (5): 519–532. doi:10.1007/s40256-023-00599-0. PMID 37526885. S2CID 260348901.
- ^ Packard, Elizabeth; de Feria, Alejandro; Peshin, Supriya; Reza, Nosheen; Owens, Anjali Tiku (December 2022). “Contemporary Therapies and Future Directions in the Management of Hypertrophic Cardiomyopathy”. Cardiology and Therapy. 11 (4): 491–507. doi:10.1007/s40119-022-00283-5. PMC 9652179.