From Wikipedia, the free encyclopedia
Content deleted Content added
|
|
|||
| Line 16: | Line 16: | ||
|
==Preparation== |
==Preparation== |
||
| ⚫ | |||
|
===History=== |
|||
| ⚫ |
A number of chemists in the early 1900s, including Staudinger and Pfenninger (1916), as well as Delepine (1920) studied episulfides.Sander, M. Thiiranes. Chem. Rev. 1966, 66(3), 297-339. {{doi|10.1021/cr60241a004}}
|
||
|
===Contemporary methods=== |
|||
|
Following the lead of Dachlauer and Jackel, contemporary routes to episulfides utilize a two-step method, converting an olefin to an epoxide followed by thiation using [[thiocyanate]] or [[thiourea]].<ref name=”Sander” /> |
|||
|
:[[Image:episulfide pic2.png|450px]] |
:[[Image:episulfide pic2.png|450px]] |
||
|
Episulfides can also be prepared from cyclic [[carbonate ester|carbonates]], [[hydroxymercaptan]]s, [[halohydrin]]s, [[dihaloalkane]]s, and [[halomercaptan]]s.<ref name=”Sander” /> For example, the reaction of [[ethylene carbonate]] and [[potassium thiocyanate|KSCN]] gives [[ethylene sulfide]]:<ref>{{cite journal| author1 = Searles, S. | author2 = Lutz, E. F. | author3 = Hays, H. R. | author4 = Mortensen, H. E. | title = Ethylene Sulfide | journal = Organic Syntheses | year = 1962| volume = 42 | page = 59 | doi= 10.15227/orgsyn.042.0059}}</ref> |
Episulfides can also be prepared from cyclic [[carbonate ester|carbonates]], [[hydroxymercaptan]]s, [[halohydrin]]s, [[dihaloalkane]]s, and [[halomercaptan]]s.<ref name=”Sander” /> For example, the reaction of [[ethylene carbonate]] and [[potassium thiocyanate|KSCN]] gives [[ethylene sulfide]]:<ref>{{cite journal| author1 = Searles, S. | author2 = Lutz, E. F. | author3 = Hays, H. R. | author4 = Mortensen, H. E. | title = Ethylene Sulfide | journal = Organic Syntheses | year = 1962| volume = 42 | page = 59 | doi= 10.15227/orgsyn.042.0059}}</ref> |
||
|
: |
:KSCN + C2H4O2CO -> KOCN + C2H4S + CO2 |
||
|
The metal-catalyzed reaction of sulfur with alkenes has been demonstrated.<ref>{{cite journal|doi=10.1021/cr030005p|title=Synthesis of Thiiranes by Direct Sulfur Transfer: The Challenge of Developing Effective Sulfur Donors and Metal Catalysts|year=2004|last1=Adam|first1=Waldemar|last2=Bargon|first2=Rainer M.|journal=Chemical Reviews|volume=104|issue=1|pages=251–262|pmid=14719976}}</ref> |
The metal-catalyzed reaction of sulfur with alkenes has been demonstrated.<ref>{{cite journal|doi=10.1021/cr030005p|title=Synthesis of Thiiranes by Direct Sulfur Transfer: The Challenge of Developing Effective Sulfur Donors and Metal Catalysts|year=2004|last1=Adam|first1=Waldemar|last2=Bargon|first2=Rainer M.|journal=Chemical Reviews|volume=104|issue=1|pages=251–262|pmid=14719976}}</ref> |
||
|
:<math>\text{alkene} + \text{S } \longrightarrow \text{ episulfide}</math> |
|||
|
Tetrasubstituted thiiranes form spontaneously from the corresponding [[sulfur ylide|thiocarbonyl ylide]].<ref>{{cite journal|author1 =Rolf Huisgen|author-link =Rolf Huisgen|author2 = J. Rapp |title=1,3-Dipolar Cycloadditions. 98. The Chemistry of Thiocarbonyl ”S”-Sulfides |journal=Tetrahedron |date=1997 |volume=53 |issue=3 |page=950 |doi=10.1016/S0040-4020(96)01068-X}}</ref> |
Tetrasubstituted thiiranes form spontaneously from the corresponding [[sulfur ylide|thiocarbonyl ylide]].<ref>{{cite journal|author1 =Rolf Huisgen|author-link =Rolf Huisgen|author2 = J. Rapp |title=1,3-Dipolar Cycloadditions. 98. The Chemistry of Thiocarbonyl ”S”-Sulfides |journal=Tetrahedron |date=1997 |volume=53 |issue=3 |page=950 |doi=10.1016/S0040-4020(96)01068-X}}</ref> |
||
Latest revision as of 14:23, 4 February 2026
Organic compounds with a saturated carbon-carbon-sulfur ring
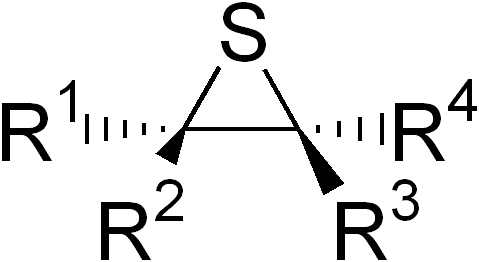
In organic chemistry, an episulfide is an organic compound that contain a saturated, heterocyclic ring consisting of two carbon atoms and one sulfur atom. It is the sulfur analogue of an epoxide or aziridine. They are also known as thiiranes, olefin sulfides, thioalkylene oxides,[citation needed] and thiacyclopropanes. Episulfides are less common and generally less stable than epoxides. The most common derivative is ethylene sulfide (C2H4S).[1][2]
According to electron diffraction, the C−C and C−S distances in ethylene sulfide are respectively 1.473 and 1.811 Å. The C−C−S and C−S−C angles are respectively 66.0 and 48.0°.[1]
A number of chemists in the early 1900s, including Staudinger and Pfenninger (1916), as well as Delepine (1920) studied episulfides. In 1934 Dachlauer and Jackel devised a general synthesis of episulfides from epoxides using alkali thiocyanates and thiourea.[3]
Episulfides can also be prepared from cyclic carbonates, hydroxymercaptans, halohydrins, dihaloalkanes, and halomercaptans.[3] For example, the reaction of ethylene carbonate and KSCN gives ethylene sulfide:[4]
- KSCN + C2H4O2CO → KOCN + C2H4S + CO2
The metal-catalyzed reaction of sulfur with alkenes has been demonstrated.[5]
Tetrasubstituted thiiranes form spontaneously from the corresponding thiocarbonyl ylide.[6]
Episulfides, due to their innate ring strain, often undergo ring-opening reactions, especially with nucleophiles.[3] For terminal episulfides, nucleophiles attack the primary carbon:
- R2C(S)CH2 + NuH → R2CHCH2SNu
Nucleophiles include hydrides, thiolates, alkoxides, amines, and carbanions.
Thiiranes occur very rarely in nature and are of no significance medicinally.[1] They are intermediate in the action of sulfur mustards, a family of chemical weapons.[7]
Very few commercial applications exist, although the polymerization of episulfide has been reported.[8]
Dithiirane is three membered ring containing two sulfur atoms and one carbon. One example was prepared by oxidation of a 1,3-dithietane.[9]
- ^ a b c Wataru Ando; Nami Choi; Norihiro Tokitoh (1996). “Thiiranes and Thiirenes: Monocyclic”. Thiiranes and Thiirenes: Monocyclic. Comprehensive Heterocyclic Chemistry II. Vol. 1A. pp. 173–240. doi:10.1016/B978-008096518-5.00005-8. ISBN 978-0-08-096518-5.
- ^ Warren Chew; David N. Harpp (1993). “Recent aspects of thiirane chemistry”. Journal of Sulfur Chemistry. 15 (1): 1–39. doi:10.1080/01961779308050628.
- ^ a b c Sander, M. Thiiranes. Chem. Rev. 1966, 66(3), 297-339. doi:10.1021/cr60241a004
- ^ Searles, S.; Lutz, E. F.; Hays, H. R.; Mortensen, H. E. (1962). “Ethylene Sulfide”. Organic Syntheses. 42: 59. doi:10.15227/orgsyn.042.0059.
- ^ Adam, Waldemar; Bargon, Rainer M. (2004). “Synthesis of Thiiranes by Direct Sulfur Transfer: The Challenge of Developing Effective Sulfur Donors and Metal Catalysts”. Chemical Reviews. 104 (1): 251–262. doi:10.1021/cr030005p. PMID 14719976.
- ^ Rolf Huisgen; J. Rapp (1997). “1,3-Dipolar Cycloadditions. 98. The Chemistry of Thiocarbonyl S-Sulfides”. Tetrahedron. 53 (3): 950. doi:10.1016/S0040-4020(96)01068-X.
- ^ Mustard agents: description, physical and chemical properties, mechanism of action, symptoms, antidotes and methods of treatment. Organisation for the Prohibition of Chemical Weapons. Accessed June 8, 2010.
- ^ Nakano, K.; Tatsumi, G.; Nazaki, K. (2007). “Synthesis of Sulfur-Rich Polymers: Copolymerization of Episulfide with Carbon Disulfide by Using [PPN]Cl/(salph)Cr(III)Cl System”. J. Am. Chem. Soc. 129 (49): 15116–15117. Bibcode:2007JAChS.12915116N. doi:10.1021/ja076056b. PMID 17999507.
- ^ a b Ahhiko Ishii; Masamatsu Hoshino; Juzo Nakayama (2009). “Recent Advances in Chemistry of Dithiirane and Small Ring Compounds Containing Two Chalcogen Atoms”. Pure Appl. Chem. 68 (4): 869–874. doi:10.1351/pac199668040869. S2CID 55817318.