From Wikipedia, the free encyclopedia
Content deleted Content added
|
Tag:
|
|
||
| Line 1: | Line 1: | ||
|
{{Dashboard.wikiedu.org draft template/about this sandbox}} |
|||
|
== Erogorgiaene == |
== Erogorgiaene == |
||
|
{{Infobox |
{{Infobox |
||
Latest revision as of 00:07, 2 December 2025
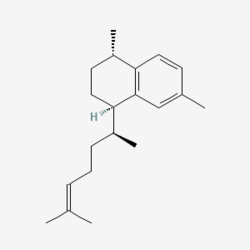
Erogorgiaene is a bicyclic compound found in marine octocoral Psuedoptergorgia elisabethae. It is used as a intermediate to synthesize pseudopterosin and is made by dehydrogenizing elisabethatriene so that a benezene ring is formed.[1]
Erogorgiaene was found to have potential as a antibiotic against mycobacterium tuberculosis.[2] However limited natural supply and lack of experimentation for human or veteranary consumption keeps it out of any current medication.
- ^ Kohl, Amber C.; Kerr, Russell G. (2003-11-26). “Pseudopterosin Biosynthesis: Aromatization of the Diterpene Cyclase Product, Elisabethatriene”. Marine Drugs. 1 (1): 54–65. doi:10.3390/md101054. ISSN 1660-3397.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ Incerti-Pradillos, Celia A.; Kabeshov, Mikhail A.; O’Hora, Paul S.; Shipilovskikh, Sergei A.; Rubtsov, Aleksandr E.; Drobkova, Vera A.; Balandina, Svetlana Yu.; Malkov, Andrei V. (2016). “Asymmetric Total Synthesis of (−)-Erogorgiaene and Its C-11 Epimer and Investigation of Their Antimycobacterial Activity”. Chemistry – A European Journal. 22 (40): 14390–14396. doi:10.1002/chem.201602440. ISSN 1521-3765.