From Wikipedia, the free encyclopedia
Content deleted Content added
| Line 74: | Line 74: | ||
|
== Development == |
== Development == |
||
|
=== Background === |
=== Background === |
||
|
Ibuzatrelvir’s design is derived from the structural framework of nirmatrelvir and exhibits extensive antiviral activity against coronaviruses in vitro, demonstrating efficacy in a mouse-adapted SARS-CoV-2 model following oral administration.<ref name=”MDPI2024″>{{cite journal |title=Inhibitors of SARS-CoV-2 Main Protease (Mpro) as Anti-Coronavirus Agents |journal=Biomolecules |date=2024-07-04 |url=https://www.mdpi.com/2218-273X/14/7/797}}</ref> Pfizer announced ibuzatrelvir as a second-generation SARS-CoV-2 M<sup>pro</sup> drug candidate, which has received fast-track status and has successfully completed Phase 2 clinical trials.<ref name=” |
Ibuzatrelvir’s design is derived from the structural framework of nirmatrelvir and exhibits extensive antiviral activity against coronaviruses in vitro, demonstrating efficacy in a mouse-adapted SARS-CoV-2 model following oral administration.<ref name=”MDPI2024″>{{cite journal |title=Inhibitors of SARS-CoV-2 Main Protease (Mpro) as Anti-Coronavirus Agents |journal=Biomolecules |date=2024-07-04 |url=https://www.mdpi.com/2218-273X/14/7/797}}</ref> Pfizer announced ibuzatrelvir as a second-generation SARS-CoV-2 M<sup>pro</sup> drug candidate, which has received fast-track status and has successfully completed Phase 2 clinical trials.<ref name=””> |
||
|
=== Advantages over Paxlovid === |
=== Advantages over Paxlovid === |
||
|
Ibuzatrelvir offers several advantages over its predecessor, particularly that it doesn’t have the drug-drug interactions that prevent many people with COVID-19 from taking Paxlovid.<ref name=”CEN2025″/><ref name=”ACS2025″>{{cite journal |title=Pfizer readies its next-generation COVID-19 antiviral |journal=C&EN Global Enterprise |url=https://pubs.acs.org/doi/10.1021/cen-10306-cover3}}</ref> It is more metabolically stable and has fewer potential drug-drug interactions.<ref name=” |
Ibuzatrelvir offers several advantages over its predecessor, particularly that it doesn’t have the drug-drug interactions that prevent many people with COVID-19 from taking Paxlovid.<ref name=”CEN2025″/><ref name=”ACS2025″>{{cite journal |title=Pfizer readies its next-generation COVID-19 antiviral |journal=C&EN Global Enterprise |url=https://pubs.acs.org/doi/10.1021/cen-10306-cover3}}</ref> It is more metabolically stable and has fewer potential drug-drug interactions.<ref name=””/><ref name=”MDPI2024″/> Additionally, participants in ibuzatrelvir’s Phase 2 clinical trial did not report “Paxlovid mouth,” the unpleasant metallic taste side effect commonly associated with Paxlovid treatment.<ref name=”CEN2025″/> |
||
|
== Clinical trials == |
== Clinical trials == |
||
| Line 86: | Line 86: | ||
|
=== Phase 3 trial === |
=== Phase 3 trial === |
||
|
Pfizer has begun Phase 3 clinical trials of ibuzatrelvir.<ref name=”CEN2025 |
Pfizer has begun Phase 3 clinical trials of ibuzatrelvir.<ref name=”CEN2025″/> The Phase 3 trial of ibuzatrelvir is expected to provide valuable insights into how this new treatment compares to existing options in reducing disease severity.<ref name=”Contagion2024″>{{cite web |title=Pfizer Launched Phase 3 Trial for Ibuzatrelvir, New Oral Antiviral for COVID-19 Treatment |url=https://www.contagionlive.com/view/pfizer-launched-phase-3-trial-for-ibuzatrelvir-a-new-oral-antiviral-for-covid-19-treatment |website=Contagion Live |date=2024-12-24 |access-date=2025-09-16}}</ref> |
||
|
== Safety profile == |
== Safety profile == |
||
|
Ibuzatrelvir is an orally bioavailable SARS-CoV-2 M<sup>pro</sup> inhibitor with demonstrated in vitro antiviral activity and low potential for safety concerns, including drug-drug interactions.<ref name=”Oxford2025″/> |
Ibuzatrelvir is an orally bioavailable SARS-CoV-2 M<sup>pro</sup> inhibitor with demonstrated in vitro antiviral activity and low potential for safety concerns, including drug-drug interactions.<ref name=”Oxford2025″/> |
||
|
== Regulatory status == |
|||
|
As of 2025, ibuzatrelvir remains an investigational drug undergoing clinical development. The compound has received fast-track status from regulatory authorities.<ref name=”PMC2024″/> |
|||
|
== References == |
== References == |
||
|
{{reflist}} |
{{reflist}} |
||
Revision as of 12:10, 16 September 2025
Antiviral drug
Pharmaceutical compound
 |
|
| Other names | PF-07817883 |
|---|---|
| Routes of administration |
Oral |
| Legal status | |
|
|
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| Formula | C21H30F3N5O5 |
| Molar mass | 489.496 g·mol−1 |
| 3D model (JSmol) | |
|
|
|
|
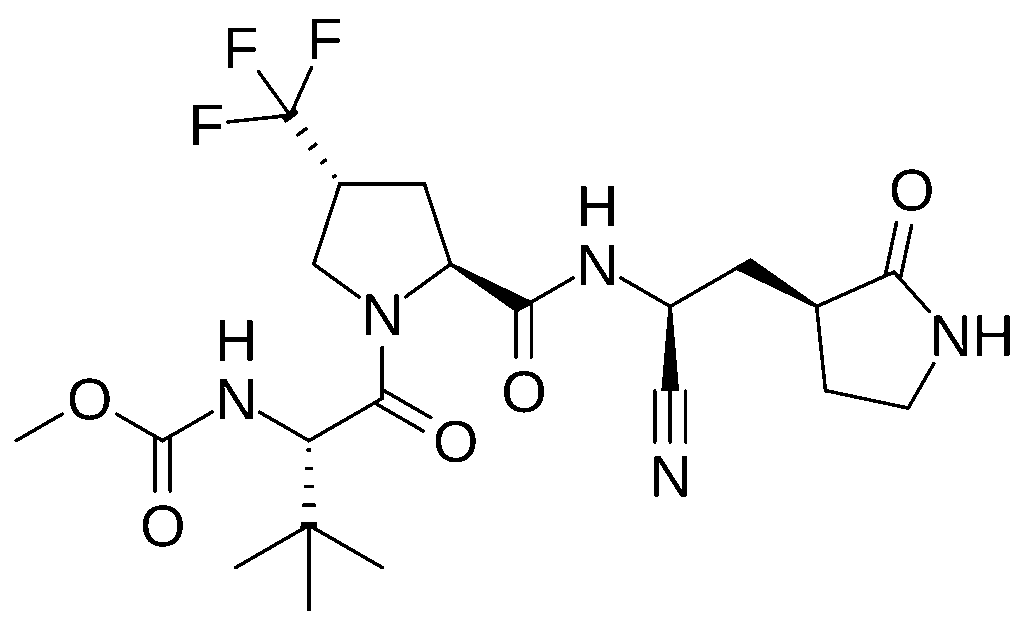
Ibuzatrelvir (also known as PF-07817883) is an investigational antiviral drug developed by Pfizer for the treatment of COVID-19.[1] It is a second-generation oral SARS-CoV-2 main protease (Mpro) inhibitor designed to improve upon the limitations of nirmatrelvir, the active antiviral component of Paxlovid, which is currently the only oral antiviral approved by the FDA for COVID-19 treatment.[2][3][4]
Mechanism of action
Ibuzatrelvir is an orally bioavailable SARS-CoV-2 main protease (Mpro) inhibitor that targets viral replication through reversible covalent interaction with the active site cysteine residue (Cys145) of the viral 3CL protease.[5][6][7] Like nirmatrelvir in Paxlovid, this orally active drug candidate is designed to target viral main proteases through reversible covalent interaction of its nitrile warhead with the active site thiol of the chymotrypsin-like cysteine protease.[8]
The drug has demonstrated pan-human coronavirus antiviral activity and favorable off-target selectivity profile in vitro and in preclinical studies.[5]
Development
Background
Ibuzatrelvir’s design is derived from the structural framework of nirmatrelvir and exhibits extensive antiviral activity against coronaviruses in vitro, demonstrating efficacy in a mouse-adapted SARS-CoV-2 model following oral administration.[9] Pfizer announced ibuzatrelvir as a second-generation SARS-CoV-2 Mpro drug candidate, which has received fast-track status and has successfully completed Phase 2 clinical trials.Cite error: A <ref> tag is missing the closing </ref> (see the help page). It is more metabolically stable and has fewer potential drug-drug interactions.[6][9] Additionally, participants in ibuzatrelvir’s Phase 2 clinical trial did not report “Paxlovid mouth,” the unpleasant metallic taste side effect commonly associated with Paxlovid treatment.[2]
Clinical trials
Phase 2b trial
A phase 2b, double-blind, randomized clinical trial enrolled US adults aged 18 to <65 years with symptomatic COVID-19 and no risk factors for severe disease. Participants were randomized 1:1:2:2 to receive 100, 300, or 600 mg ibuzatrelvir or placebo orally twice daily for 5 days.[10][11]
The study involved 240 participants with COVID-19 and demonstrated that ibuzatrelvir led to greater viral load reduction compared with placebo. All 3 tested doses were generally well tolerated.[12]
Phase 3 trial
Pfizer has begun Phase 3 clinical trials of ibuzatrelvir.[2] The Phase 3 trial of ibuzatrelvir is expected to provide valuable insights into how this new treatment compares to existing options in reducing disease severity.[13]
Safety profile
Ibuzatrelvir is an orally bioavailable SARS-CoV-2 Mpro inhibitor with demonstrated in vitro antiviral activity and low potential for safety concerns, including drug-drug interactions.[11]
Regulatory status
As of 2025, ibuzatrelvir remains an investigational drug undergoing clinical development. The compound has received fast-track status from regulatory authorities.[6]
References
- ^ Allerton CM, Arcari JT, Aschenbrenner LM, Avery M, Bechle BM, Behzadi MA, et al. (August 2024). “A Second-Generation Oral SARS-CoV-2 Main Protease Inhibitor Clinical Candidate for the Treatment of COVID-19”. Journal of Medicinal Chemistry. 67 (16): 13550–13571. doi:10.1021/acs.jmedchem.3c02469. PMC 11345836. PMID 38687966.
- ^ a b c “Pfizer readies its next-generation COVID-19 antiviral”. Chemical & Engineering News. 28 June 2024.
- ^ Chen P, Van Oers TJ, Arutyunova E, Fischer C, Wang C, Lamer T, et al. (August 2024). “A Structural Comparison of Oral SARS-CoV-2 Drug Candidate Ibuzatrelvir Complexed with the Main Protease (Mpro) of SARS-CoV-2 and MERS-CoV”. JACS Au. 4 (8): 3217–3227. doi:10.1021/jacsau.4c00508. PMC 11350714. PMID 39211604.
- ^ Brewitz L, Schofield CJ (July 2024). “Fixing the Achilles Heel of Pfizer’s Paxlovid for COVID-19 Treatment”. Journal of Medicinal Chemistry. 67 (14): 11656–11661. doi:10.1021/acs.jmedchem.4c01342. PMC 11284777. PMID 38967233.
- ^ a b “Ibuzatrelvir (PF-07817883)”. MedChemExpress. Retrieved 16 September 2025.
- ^ a b c “A Structural Comparison of Oral SARS-CoV-2 Drug Candidate Ibuzatrelvir Complexed with the Main Protease (Mpro) of SARS-CoV-2 and MERS-CoV”. JACS Au. Retrieved 16 September 2025.
- ^ “Fixing the Achilles Heel of Pfizer’s Paxlovid for COVID-19 Treatment”. Journal of Medicinal Chemistry. 25 July 2024. PMID 38967233.
- ^ “A Structural Comparison of Oral SARS-CoV-2 Drug Candidate Ibuzatrelvir Complexed with the Main Protease (Mpro) of SARS-CoV-2 and MERS-CoV”. JACS Au. Retrieved 16 September 2025.
- ^ a b “Inhibitors of SARS-CoV-2 Main Protease (Mpro) as Anti-Coronavirus Agents”. Biomolecules. 4 July 2024.
- ^ “Virologic Response and Safety of Ibuzatrelvir, A Novel SARS-CoV-2 Antiviral, in Adults With COVID-19”. PubMed. 17 March 2025. PMID 39486089.
- ^ a b “Virologic Response and Safety of Ibuzatrelvir, A Novel SARS-CoV-2 Antiviral, in Adults With COVID-19”. Clinical Infectious Diseases. 80 (3): 673. 17 March 2025.
- ^ “Virologic Response and Safety of Ibuzatrelvir, A Novel SARS-CoV-2 Antiviral, in Adults With COVID-19”. PMC. Retrieved 16 September 2025.
- ^ “Pfizer Launched Phase 3 Trial for Ibuzatrelvir, New Oral Antiviral for COVID-19 Treatment”. Contagion Live. 24 December 2024. Retrieved 16 September 2025.