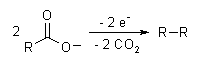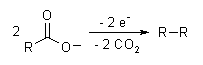From Wikipedia, the free encyclopedia
Content deleted Content added
|
|
|||
| Line 27: | Line 27: | ||
|
: R<sub>1</sub>COO<sup>−</sup> + R<sub>2</sub>COO<sup>−</sup> → R<sub>1</sub>−R<sub>1</sub> and/or R<sub>1</sub>−R<sub>2</sub> and/or R<sub>2</sub>−R<sub>2</sub> |
: R<sub>1</sub>COO<sup>−</sup> + R<sub>2</sub>COO<sup>−</sup> → R<sub>1</sub>−R<sub>1</sub> and/or R<sub>1</sub>−R<sub>2</sub> and/or R<sub>2</sub>−R<sub>2</sub> |
||
|
|
can the reaction the in protic ], and .<ref name=Encyc>{{cite encyclopedia|doi=10.1002/9781119953678.rad034|at=§ 2.7|=Electrochemically initiated radical reactions|volume=Synthetic Strategies and Applications|first1=Martin A.|=Bohn|first2=Anna|last2=Paul|first3=Gerhard|last3=Hilt|date=15 March 2012|publisher=Wiley}}</ref> |
||
|
In 2022, it was discovered that the Kolbe electrolysis is enhanced if an alternating square wave current is used instead of a [[direct current]].<ref>{{Cite report |url=https://chemrxiv.org/engage/chemrxiv/article-details/635afdb2ca86b81d1ac683f4 |title=Overcoming the Limitations of Kolbe Coupling via Waveform-Controlled Electrosynthesis |last1=Hioki |first1=Yuta |last2=Costantini |first2=Matteo |date=2022-10-31 |publisher=Chemistry |doi=10.26434/chemrxiv-2022-3cj82-v2 |last3=Griffin |first3=Jeremy |last4=Harper |first4=Kaid |last5=Prado Merini |first5=Melania |last6=Nissl |first6=Benedikt |last7=Kawamata |first7=Yu |last8=Baran |first8=Phil|doi-access=free }}</ref><ref>{{Cite journal |last1=Hioki |first1=Yuta |last2=Costantini |first2=Matteo |last3=Griffin |first3=Jeremy |last4=Harper |first4=Kaid C. |last5=Merini |first5=Melania Prado |last6=Nissl |first6=Benedikt |last7=Kawamata |first7=Yu |last8=Baran |first8=Phil S. |date=2023-04-07 |title=Overcoming the limitations of Kolbe coupling with waveform-controlled electrosynthesis |url=https://www.science.org/doi/10.1126/science.adf4762 |journal=Science |language=en |volume=380 |issue=6640 |pages=81–87 |doi=10.1126/science.adf4762 |pmid=37023204 |issn=0036-8075|url-access=subscription }}</ref> |
In 2022, it was discovered that the Kolbe electrolysis is enhanced if an alternating square wave current is used instead of a [[direct current]].<ref>{{Cite report |url=https://chemrxiv.org/engage/chemrxiv/article-details/635afdb2ca86b81d1ac683f4 |title=Overcoming the Limitations of Kolbe Coupling via Waveform-Controlled Electrosynthesis |last1=Hioki |first1=Yuta |last2=Costantini |first2=Matteo |date=2022-10-31 |publisher=Chemistry |doi=10.26434/chemrxiv-2022-3cj82-v2 |last3=Griffin |first3=Jeremy |last4=Harper |first4=Kaid |last5=Prado Merini |first5=Melania |last6=Nissl |first6=Benedikt |last7=Kawamata |first7=Yu |last8=Baran |first8=Phil|doi-access=free }}</ref><ref>{{Cite journal |last1=Hioki |first1=Yuta |last2=Costantini |first2=Matteo |last3=Griffin |first3=Jeremy |last4=Harper |first4=Kaid C. |last5=Merini |first5=Melania Prado |last6=Nissl |first6=Benedikt |last7=Kawamata |first7=Yu |last8=Baran |first8=Phil S. |date=2023-04-07 |title=Overcoming the limitations of Kolbe coupling with waveform-controlled electrosynthesis |url=https://www.science.org/doi/10.1126/science.adf4762 |journal=Science |language=en |volume=380 |issue=6640 |pages=81–87 |doi=10.1126/science.adf4762 |pmid=37023204 |issn=0036-8075|url-access=subscription }}</ref> |
||
|
=== Hofer–Moest reaction === |
|||
|
In the ”’Hofer–Moest reaction”’, the alkyl radical undergo further oxidation to form a carbocation, rather than coupling with another alkyl radical, which then reacts with an available [[nucleophile]].<ref>{{cite book |title= Comprehensive Organic Name Reactions and Reagents |chapter= Hofer-Moest Reaction |date= 2010 |pages= 1443–1446 |doi= 10.1002/9780470638859.conrr322 |isbn= 978-0-471-70450-8 }}</ref> The Hofer-Moest reaction always occurs if the carboxylic acid bears a carbocation-stabilizing side-substituent at the α position, but only sometimes otherwise.<ref name=Encyc/> |
|||
|
==Applications== |
==Applications== |
||
Latest revision as of 01:53, 12 November 2025
Organic reaction
The Kolbe electrolysis or Kolbe reaction is an organic reaction named after Hermann Kolbe.[1] The Kolbe reaction is formally a decarboxylative dimerisation of two carboxylic acids (or carboxylate ions). The overall reaction is:
Mechanism and side-reactions
[edit]
The reaction mechanism involves a two-stage radical process: electrochemical oxidation first gives a alkylcarboxyl radical, which decarboxylates almost immediately to give an alkyl radical intermediate. The alkyl radicals which combine to form a covalent bond.[2] As an example, electrolysis of acetic acid yields ethane and carbon dioxide:
- CH3COOH → CH3COO− → CH3COO· → CH3· + CO2
- 2CH3· → CH3CH3
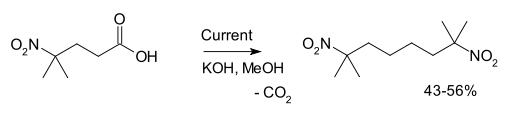
Another example is the synthesis of 2,7-dimethyl-2,7-dinitrooctane from 4-methyl-4-nitrovaleric acid:[3]
Other compounds can trap the radicals formed by decarboxylation, and the Kolbe reaction has also been occasionally used in cross-coupling reactions.
If a mixture of two different carboxylates are used, the radical cross-coupling reaction generally gives all combinations of them:[4]
- R1COO− + R2COO− → R1−R1 and/or R1−R2 and/or R2−R2
The reaction process can be enhanced and the Hofer–Moest reaction alternative supressed, by performing the reaction under weakly acidic conditions in protic solvents, and using a high curent density and a platinum anodic electrode.[4]
In 2022, it was discovered that the Kolbe electrolysis is enhanced if an alternating square wave current is used instead of a direct current.[5][6]
Hofer–Moest reaction
[edit]
In the Hofer–Moest reaction, the alkyl radical undergo further oxidation to form a carbocation, rather than coupling with another alkyl radical, which then reacts with an available nucleophile.[7] The Hofer-Moest reaction always occurs if the carboxylic acid bears a carbocation-stabilizing side-substituent at the α position, but only sometimes otherwise.[4]
Kolbe electrolysis has a few industrial applications.[8] The reaction typically yields <50%.[4]
In one example, sebacic acid has been produced commercially by Kolbe electrolysis of adipic acid.[9]
Kolbe electrolysis has been examined for converting biomass into biodiesel[10][11] and for grafting of carbon electrodes.[12][13]
- ^ Utley, James (1997). “Trends in Organic Electrosynthesis”. Chemical Society Reviews. 26 (3): 157. doi:10.1039/cs9972600157.
- ^ Vijh, A. K.; Conway, B. E. (1967). “Electrode Kinetic Aspects of the Kolbe Reaction”. Chem Rev. 67 (6): 623–664. doi:10.1021/cr60250a003.
- ^ Sharkey, W. H.; Langkammerer, C. M. (1973). “2,7-Dimethyl-2,7-dinitrooctane”. Organic Syntheses; Collected Volumes, vol. 5, p. 445.
- ^ a b c d Bohn, Martin A.; Paul, Anna; Hilt, Gerhard (15 March 2012). “Electrochemically initiated radical reactions”. Encyclopedia of Radicals in Chemistry, Biology and Materials. Vol. Synthetic Strategies and Applications. Wiley. § 2.7. doi:10.1002/9781119953678.rad034. ISBN 978-0-470-97125-3.
- ^ Hioki, Yuta; Costantini, Matteo; Griffin, Jeremy; Harper, Kaid; Prado Merini, Melania; Nissl, Benedikt; Kawamata, Yu; Baran, Phil (31 October 2022). Overcoming the Limitations of Kolbe Coupling via Waveform-Controlled Electrosynthesis (Report). Chemistry. doi:10.26434/chemrxiv-2022-3cj82-v2.
- ^ Hioki, Yuta; Costantini, Matteo; Griffin, Jeremy; Harper, Kaid C.; Merini, Melania Prado; Nissl, Benedikt; Kawamata, Yu; Baran, Phil S. (7 April 2023). “Overcoming the limitations of Kolbe coupling with waveform-controlled electrosynthesis”. Science. 380 (6640): 81–87. Bibcode:2023Sci…380…81H. doi:10.1126/science.adf4762. ISSN 0036-8075. PMID 37023204.
- ^ “Hofer-Moest Reaction”. Comprehensive Organic Name Reactions and Reagents. 2010. pp. 1443–1446. doi:10.1002/9780470638859.conrr322. ISBN 978-0-471-70450-8.
- ^ Wendt, Hartmut; Vogt, Helmut; Kreysa, Gerhard; m. Kolb, Dieter; e. Engelmann, Gerald; Ziegler, Jörg C.; Goldacker, Hubert; Jüttner, Klaus; Galla, Ulrich; Schmieder, Helmut; Steckhan, Eberhard (2009). “Electrochemistry”. Ullmann’s Encyclopedia of Industrial Chemistry. doi:10.1002/14356007.a09_183.pub3. ISBN 978-3-527-30673-2.
- ^ Seko, Maomi; Yomiyama, Akira; Isoya, Toshiro (1979). “Development of Kolbe Electrosynthesis of Sebacic Acid”. CEER, Chemical Economy & Engineering Review. 11 (9): 48–50.
- ^ Yuan, Gang; Wu, Chan; Zeng, Guorong; Niu, Xiaopo; Shen, Guoqiang; Wang, Li; Zhang, Xiangwen; Luque, Rafael; Wang, Qingfa (18 January 2020). “Kolbe Electrolysis of Biomass-Derived Fatty Acids Over Pt Nanocrystals in an Electrochemical Cell”. ChemCatChem. 12 (2): 642–648. doi:10.1002/cctc.201901443. ISSN 1867-3880.
- ^ Ho, Calvin K.; McAuley, Kimberley B.; Peppley, Brant A. (1 October 2019). “Biolubricants through renewable hydrocarbons: A perspective for new opportunities”. Renewable and Sustainable Energy Reviews. 113 109261. doi:10.1016/j.rser.2019.109261. ISSN 1364-0321.
- ^ Andrieux, Claude P.; Gonzalez, Felipe; Savéant, Jean-Michel (1 May 1997). “Derivatization of Carbon Surfaces by Anodic Oxidation of Arylacetates. Electrochemical Manipulation of the Grafted Films”. Journal of the American Chemical Society. 119 (18): 4292–4300. doi:10.1021/ja9636092. ISSN 0002-7863.
- ^ Bélanger, Daniel; Pinson, Jean (20 June 2011). “Electrografting: a powerful method for surface modification”. Chemical Society Reviews. 40 (7): 3995–4048. doi:10.1039/C0CS00149J. ISSN 1460-4744. PMID 21503288.