As can be seen nearly all of the Ki activity resides in a single optical antipode (or enantiomer).
As can be seen nearly all of the Ki activity resides in a single optical antipode (or enantiomer).
While McN-4612-Y does not behave as a positive reinforcer, according to the reference it might possibly have utility as an antipsychotic.<ref>{{cite journal | vauthors=((Chrzanowski, F. A.)), ((McGrogan, B. A.)), ((Maryanoff, B. E.)) | journal=Journal of Medicinal Chemistry | title=The pKa of butaclamol and the mode of butaclamol binding to central dopamine receptors | volume=28 | issue=3 | pages=399–400 | date= March 1985 | url=https://pubs.acs.org/doi/abs/10.1021/jm00381a022 | doi=10.1021/jm00381a022}}</ref>
While McN-4612-Y does not as a positive reinforcer, according to the reference it might possibly have utility as an antipsychotic.<ref>{{cite journal | vauthors=((Chrzanowski, F. A.)), ((McGrogan, B. A.)), ((Maryanoff, B. E.)) | journal=Journal of Medicinal Chemistry | title=The pKa of butaclamol and the mode of butaclamol binding to central dopamine receptors | volume=28 | issue=3 | pages=399–400 | date= March 1985 | url=https://pubs.acs.org/doi/abs/10.1021/jm00381a022 | doi=10.1021/jm00381a022}}</ref>
==Self-administration studies==
==Self-administration studies==
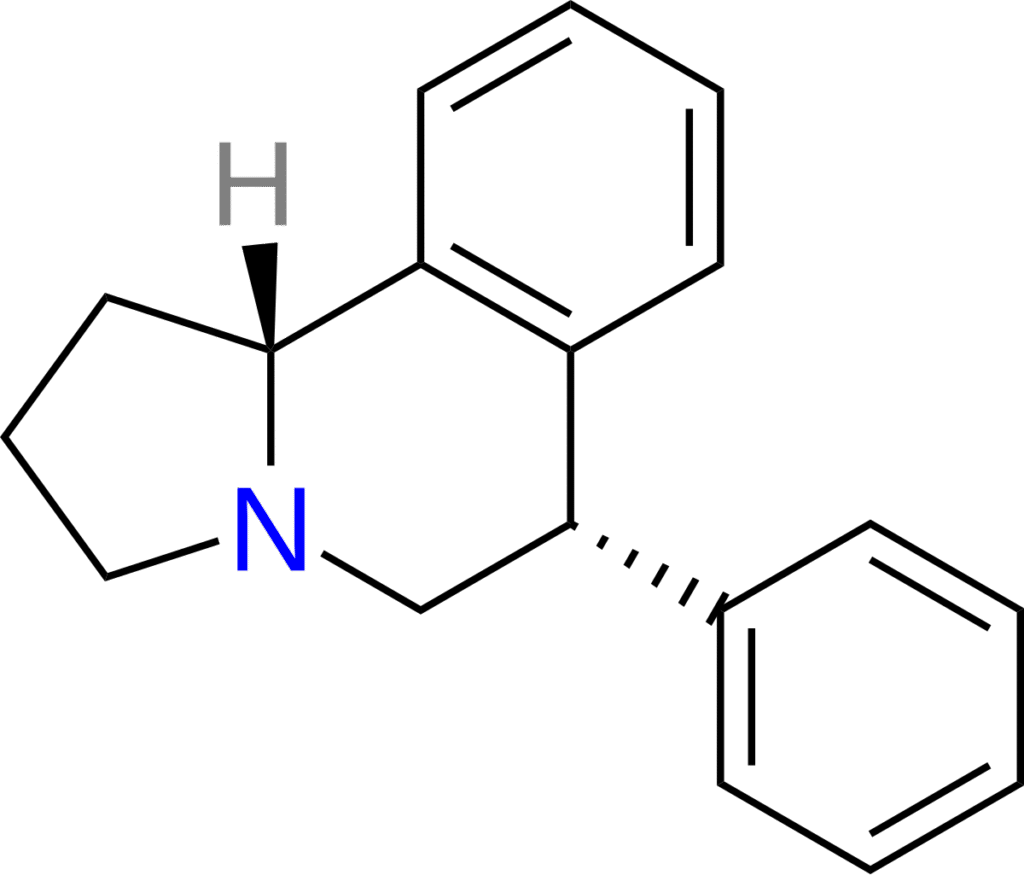
Pharmaceutical compound
| Other names | Mcn 4612 |
|---|---|
|
|
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| Formula | C18H19N |
| Molar mass | 249.357 g·mol−1 |
| 3D model (JSmol) | |
|
|
|
|
McN-4612 is a noradrenaline preferring SNDRI (aka TRI) developed by McNeil Laboratories in the 1980’s. [1]
The Ki digits are reported to be the following (nM):
| Compound | NE | DA | 5-HT |
|---|---|---|---|
| racemic | 0.6 | 11.3 | 23.5 |
| McN 4612-z | 0.37 | 4.4 | 12.4 |
| McN-4612-Y | 411 | 1345 | 4009 |
As can be seen nearly all of the Ki activity resides in a single optical antipode (or enantiomer).
While McN-4612-Y does not function as a positive reinforcer, according to the reference it might possibly have utility as an antipsychotic.[2]
Self-administration studies
[edit]
Although McN-4612 was never publically taken by humans, the self-administration by rats is reported in the literature.[3]
McN-4612 is the lead compound in a series of agents and the antecedent to such agents as McN5652, McN 5707, JNJ-7925476, Mcn-5292, McN-5558, McN-5908 or McN-5847 for example. The SAR can be manipulated to place particular emphasis on catecholaminergic psychostimulants or to incorporate 5-HT into the pharmacophore. Some of these compounds are tremendously powerful agents. At the time of discovery, McN-5908 was the most powerful psychostimulant ever discovered, although it was claimed that it was also quite toxic. Only a few of the phenyltropanes are believed to have potencies that can rival this agent.
A number of methods are available to synthesizing these agents in the appendant literature.
The first scheme relies on 2,2-Diphenylethylamine [3963-62-0], which itself is the product of the reduction of diphenylacetonitrile.
However, the 2-phenylpyrrolidine [1006-64-0] route is also attractive. Recently, an enantiomerically selective method was reported using this method of synthesis.[4]
- ^ Maryanoff BE, McComsey DF, Castanzo MJ, Setler PE, Gardocki JF, Shank RP, Schneider CR (August 1984). “Pyrroloisoquinoline antidepressants. Potent, enantioselective inhibition of tetrabenazine-induced ptosis and neuronal uptake of norepinephrine, dopamine, and serotonin”. Journal of Medicinal Chemistry. 27 (8): 943–6. doi:10.1021/jm00374a001. PMID 6747993.
- ^ Chrzanowski, F. A., McGrogan, B. A., Maryanoff, B. E. (March 1985). “The pKa of butaclamol and the mode of butaclamol binding to central dopamine receptors”. Journal of Medicinal Chemistry. 28 (3): 399–400. doi:10.1021/jm00381a022.
- ^ Nielsen, J., Duda, N., Mokler, D., Moore, K. (February 1984). “Self-administration of central stimulants by rats: A comparison of the effects of d-amphetamine, methylphenidate and McNeil 4612”. Pharmacology Biochemistry and Behavior. 20 (2): 227–232. doi:10.1016/0091-3057(84)90247-8.
- ^ Zhang, Y., Kong, D., Wang, R., Hou, G. (2017). “Synthesis of chiral cyclic amines via Ir-catalyzed enantioselective hydrogenation of cyclic imines”. Organic & Biomolecular Chemistry. 15 (14): 3006–3012. doi:10.1039/C7OB00442G.