Alpha-synuclein has a [[Prion|prion-like]] molecular spread and is suggested to be released through rare exocytosis pathways.<ref name=”:2″ /> This release with [[Exosome (vesicle)|exosomes]] on their way to degradation in [[Lysosome|lysosomes]] suggest this process may be calcium-dependent, and therefore suggests propagation of misfolded alpha-synuclein between neurons synaptically connected.<ref name=”:2″ /> Neuronal death caused by aggregated alpha-synuclein may also further accelerate the formation of these toxic aggregates, which can then trigger a selective progression of neuronal death through impairment of the [[Mitochondrion|mitochondria]], alteration of [[Calcium metabolism|calcium homeostasis]], and lysosomal dysfunction. <ref name=”:12″>{{Cite journal |last=Yaribash |first=Shakila |last2=Mohammadi |first2=Keyhan |last3=Sani |first3=Mahmood Alizadeh |date=2025-03-26 |title=Alpha-Synuclein Pathophysiology in Neurodegenerative Disorders: A Review Focusing on Molecular Mechanisms and Treatment Advances in Parkinson’s Disease |url=https://link.springer.com/10.1007/s10571-025-01544-2 |journal=Cellular and Molecular Neurobiology |language=en |volume=45 |issue=1 |doi=10.1007/s10571-025-01544-2 |issn=1573-6830 |pmc=11947388 |pmid=40140103}}</ref><ref name=”:3″>{{Cite journal |last=Calabresi |first=Paolo |last2=Mechelli |first2=Alessandro |last3=Natale |first3=Giuseppina |last4=Volpicelli-Daley |first4=Laura |last5=Di Lazzaro |first5=Giulia |last6=Ghiglieri |first6=Veronica |date=2023-03-01 |title=Alpha-synuclein in Parkinson’s disease and other synucleinopathies: from overt neurodegeneration back to early synaptic dysfunction |url=https://www.nature.com/articles/s41419-023-05672-9 |journal=Cell Death & Disease |language=en |volume=14 |issue=3 |pages=176 |doi=10.1038/s41419-023-05672-9 |issn=2041-4889}}</ref>
Alpha-synuclein has a [[Prion|prion-like]] molecular spread and is suggested to be released through rare exocytosis pathways.<ref name=”:2″ /> This release with [[Exosome (vesicle)|exosomes]] on their way to degradation in [[Lysosome|lysosomes]] suggest this process may be calcium-dependent, and therefore suggests propagation of misfolded alpha-synuclein between neurons synaptically connected.<ref name=”:2″ /> Neuronal death caused by aggregated alpha-synuclein may also further accelerate the formation of these toxic aggregates, which can then trigger a selective progression of neuronal death through impairment of the [[Mitochondrion|mitochondria]], alteration of [[Calcium metabolism|calcium homeostasis]], and lysosomal dysfunction. <ref name=”:12″>{{Cite journal |last=Yaribash |first=Shakila |last2=Mohammadi |first2=Keyhan |last3=Sani |first3=Mahmood Alizadeh |date=2025-03-26 |title=Alpha-Synuclein Pathophysiology in Neurodegenerative Disorders: A Review Focusing on Molecular Mechanisms and Treatment Advances in Parkinson’s Disease |url=https://link.springer.com/10.1007/s10571-025-01544-2 |journal=Cellular and Molecular Neurobiology |language=en |volume=45 |issue=1 |doi=10.1007/s10571-025-01544-2 |issn=1573-6830 |pmc=11947388 |pmid=40140103}}</ref><ref name=”:3″>{{Cite journal |last=Calabresi |first=Paolo |last2=Mechelli |first2=Alessandro |last3=Natale |first3=Giuseppina |last4=Volpicelli-Daley |first4=Laura |last5=Di Lazzaro |first5=Giulia |last6=Ghiglieri |first6=Veronica |date=2023-03-01 |title=Alpha-synuclein in Parkinson’s disease and other synucleinopathies: from overt neurodegeneration back to early synaptic dysfunction |url=https://www.nature.com/articles/s41419-023-05672-9 |journal=Cell Death & Disease |language=en |volume=14 |issue=3 |pages=176 |doi=10.1038/s41419-023-05672-9 |issn=2041-4889}}</ref>
Early synaptic and [[Neuroplasticity|plastic]] alterations mediated by alpha-synuclein, as well as the mechanisms of inflammation and synaptic dysfunction that occurs before neurodegeneration, are of key interest for investigating possible therapies for synucleinpathies.<ref name=”:32″>{{Cite journal |last=Calabresi |first=Paolo |last2=Mechelli |first2=Alessandro |last3=Natale |first3=Giuseppina |last4=Volpicelli-Daley |first4=Laura |last5=Di Lazzaro |first5=Giulia |last6=Ghiglieri |first6=Veronica |date=2023-03-01 |title=Alpha-synuclein in Parkinson’s disease and other synucleinopathies: from overt neurodegeneration back to early synaptic dysfunction |url=https://www.nature.com/articles/s41419-023-05672-9 |journal=Cell Death & Disease |language=en |volume=14 |issue=3 |pages=176 |doi=10.1038/s41419-023-05672-9 |issn=2041-4889}}</ref>
==Diagnosis==
==Diagnosis==
Medical condition
| Synucleinopathy | |
|---|---|
| Other names | α-Synucleinopathies |
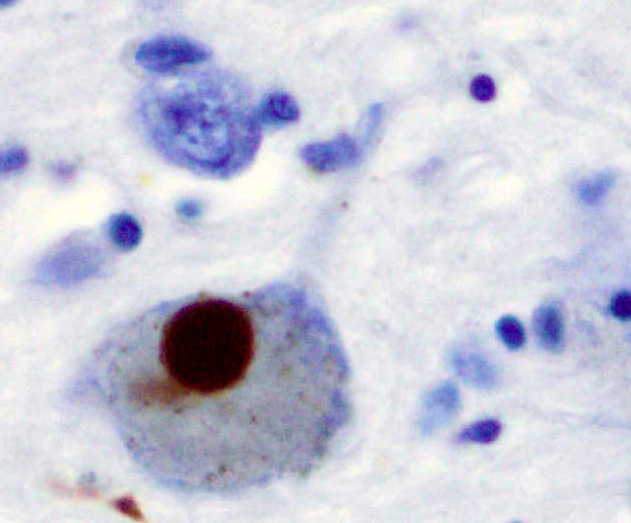
| Positive α-Synuclein staining of a Lewy body in a patient with Parkinson’s disease. | |
| Specialty | Neurology |
Synucleinopathies (also called α-Synucleinopathies) are neurodegenerative diseases characterised by the abnormal accumulation of aggregates of alpha-synuclein protein in neurons, nerve fibres or glial cells.[1] The synucleinopathies include Parkinson’s disease (PD), dementia with Lewy bodies (DLB), and multiple system atrophy (MSA).[1] Other rare disorders, such as various neuroaxonal dystrophies, also have α-synuclein pathologies.[2]
The synucleinopathies have shared features of parkinsonism, impaired cognition, sleep disorders, and visual hallucinations.[3]
Synucleinopathies can overlap with tauopathies, possibly because of interaction between the synuclein and tau proteins.[4]
REM sleep behavior disorder (RBD) is a parasomnia in which individuals with RBD lose the paralysis of muscles (atonia) that is normal during rapid eye movement (REM) sleep, and act out their dreams or have other abnormal movements or vocalizations.[5] Abnormal sleep behaviors may appear decades before any other symptoms, often as an early sign of a synucleinopathy.[6] On autopsy, 94 to 98% of individuals with polysomnography-confirmed RBD are found to have a synucleinopathy—most commonly DLB or PD.[5][7][8] Other symptoms of the specific synucleinopathy usually manifest within 15 years of the diagnosis of RBD,[9] but may emerge up to 50 years after RBD diagnosis.[5]
Alpha-synuclein deposits can affect the cardiac muscle and blood vessels.[10] Almost all people with synucleinopathies have cardiovascular dysfunction, although most are asymptomatic.[10]
From chewing to defecation, alpha-synuclein deposits affect every level of gastrointestinal function. Symptoms include upper gastrointestinal tract dysfunction such as delayed gastric emptying or lower gastrointestinal dysfunction, such as constipation and prolonged stool transit time.[10]
Urinary retention, waking at night to urinate, increased urinary frequency and urgency, and over- or underactive bladder are common in people with synucleinopathies.[10] Sexual dysfunction usually appears early in synucleinopathies, and may include erectile dysfunction, and difficulties achieving orgasm or ejaculating.[10]
The pathological aggregation of alpha-synuclein plays a key role in neurodegenerative disease.[11] The misfolding and aggregation of alpha-synuclein form toxic fibrils, which in turn form pathological inclusions, such as Lewy bodies.[12] These protein deposits are a pathological hallmark of synucleinopathies, and may interrupt crucial neuronal processes, such as functions of synaptic vesicles, leading to neuronal death.[13] Alpha-synuclein is encoded by the SNCA gene, and a mutation in this gene can lead to dysfunctions of the protein structure.[14]
Post-translational modifications are also implicated in the aggregation of alpha-synuclein, mostly occurring in the C-terminus. Phosphorylation, acetylation, ubiquitination, oxidation, and other modifications alter the structure and charge of alpha-synuclein, which can in turn lead to the formation of Lewy bodies.[15]
Alpha-synuclein has a prion-like molecular spread and is suggested to be released through rare exocytosis pathways.[15] This release with exosomes on their way to degradation in lysosomes suggest this process may be calcium-dependent, and therefore suggests propagation of misfolded alpha-synuclein between neurons synaptically connected.[15] Neuronal death caused by aggregated alpha-synuclein may also further accelerate the formation of these toxic aggregates, which can then trigger a selective progression of neuronal death through impairment of the mitochondria, alteration of calcium homeostasis, and lysosomal dysfunction. [16][17]
Early synaptic and plastic alterations mediated by alpha-synuclein, as well as the mechanisms of inflammation and synaptic dysfunction that occurs before neurodegeneration, are of key interest for investigating possible therapies for synucleinpathies.[18]
Differential diagnosis
[edit]
Persons with PD are typically less caught up in their visual hallucinations than those with DLB.[19] There is a lower incidence of tremor at rest in DLB than in PD, and signs of parkinsonism in DLB are more symmetrical.[6] In MSA, autonomic dysfunction appears earlier and is more severe, and is accompanied by uncoordinated movements, while visual hallucinations and fluctuating cognition are less common than in DLB.[20] Urinary difficulties are one of the earliest symptoms with MSA, and are often severe.[10]
- ^ a b Miglis MG, Adler CH, Antelmi E, et al. (August 2021). “Biomarkers of conversion to α-synucleinopathy in isolated rapid-eye-movement sleep behaviour disorder”. Lancet Neurol. 20 (8): 671–684. doi:10.1016/S1474-4422(21)00176-9. PMC 8600613. PMID 34302789.
- ^ Goedert M, Jakes R, Spillantini MG (2017). “The Synucleinopathies: Twenty Years On”. J Parkinsons Dis. 7 (s1): S53 – S71. doi:10.3233/JPD-179005. PMC 5345650. PMID 28282814.
- ^ Pezzoli S, Cagnin A, Bandmann O, Venneri A (July 2017). “Structural and Functional Neuroimaging of Visual Hallucinations in Lewy Body Disease: A Systematic Literature Review”. Brain Sci (Review). 7 (12): 84. doi:10.3390/brainsci7070084. PMC 5532597. PMID 28714891.
- ^ Li, Wen; Li, Jia-Yi (2024-03-26). “Overlaps and divergences between tauopathies and synucleinopathies: a duet of neurodegeneration”. Translational Neurodegeneration. 13 (1): 16. doi:10.1186/s40035-024-00407-y. ISSN 2047-9158. PMC 10964635. PMID 38528629.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ a b c St Louis EK, Boeve BF (November 2017). “REM sleep behavior disorder: Diagnosis, clinical implications, and future directions”. Mayo Clin. Proc. (Review). 92 (11): 1723–36. doi:10.1016/j.mayocp.2017.09.007. PMC 6095693. PMID 29101940.
- ^ a b St Louis EK, Boeve AR, Boeve BF (May 2017). “REM sleep behavior disorder in Parkinson’s disease and other synucleinopathies”. Mov. Disord. (Review). 32 (5): 645–58. doi:10.1002/mds.27018. PMID 28513079. S2CID 46881921.
- ^ Boot BP, McDade EM, McGinnis SM, Boeve BF (December 2013). “Treatment of dementia with Lewy bodies”. Curr Treat Options Neurol (Review). 15 (6): 738–64. doi:10.1007/s11940-013-0261-6. PMC 3913181. PMID 24222315.
- ^ Boot BP (2015). “Comprehensive treatment of dementia with Lewy bodies”. Alzheimers Res Ther (Review). 7 (1): 45. doi:10.1186/s13195-015-0128-z. PMC 4448151. PMID 26029267.
- ^ Walker Z, Possin KL, Boeve BF, Aarsland D (October 2015). “Lewy body dementias”. Lancet (Review). 386 (10004): 1683–97. doi:10.1016/S0140-6736(15)00462-6. PMC 5792067. PMID 26595642.
- ^ a b c d e f Palma JA, Kaufmann H (March 2018). “Treatment of autonomic dysfunction in Parkinson disease and other synucleinopathies”. Mov. Disord. (Review). 33 (3): 372–90. doi:10.1002/mds.27344. PMC 5844369. PMID 29508455.
- ^ Arias-Carrión, Oscar; Guerra-Crespo, Magdalena; Padilla-Godínez, Francisco J.; Soto-Rojas, Luis O.; Manjarrez, Elías (2025-06-04). “α-Synuclein Pathology in Synucleinopathies: Mechanisms, Biomarkers, and Therapeutic Challenges”. International Journal of Molecular Sciences. 26 (11): 5405. doi:10.3390/ijms26115405. ISSN 1422-0067. PMC 12155115. PMID 40508212.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ Negi, Samir; Khurana, Navneet; Duggal, Navneet (2024-07-01). “The misfolding mystery: α-synuclein and the pathogenesis of Parkinson’s disease”. Neurochemistry International. 177: 105760. doi:10.1016/j.neuint.2024.105760. ISSN 0197-0186.
{{cite journal}}: CS1 maint: article number as page number (link) - ^ Arias-Carrión, Oscar; Guerra-Crespo, Magdalena; Padilla-Godínez, Francisco J.; Soto-Rojas, Luis O.; Manjarrez, Elías (2025-06-04). “α-Synuclein Pathology in Synucleinopathies: Mechanisms, Biomarkers, and Therapeutic Challenges”. International Journal of Molecular Sciences. 26 (11): 5405. doi:10.3390/ijms26115405. ISSN 1422-0067. PMC 12155115. PMID 40508212.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ Yaribash, Shakila; Mohammadi, Keyhan; Sani, Mahmood Alizadeh (2025-03-26). “Alpha-Synuclein Pathophysiology in Neurodegenerative Disorders: A Review Focusing on Molecular Mechanisms and Treatment Advances in Parkinson’s Disease”. Cellular and Molecular Neurobiology. 45 (1). doi:10.1007/s10571-025-01544-2. ISSN 1573-6830. PMC 11947388. PMID 40140103.
- ^ a b c Burré, Jacqueline; Sharma, Manu; Südhof, Thomas C. (2018-03-01). “Cell Biology and Pathophysiology of α-Synuclein”. Cold Spring Harbor Perspectives in Medicine. 8 (3): a024091. doi:10.1101/cshperspect.a024091. ISSN 2157-1422. PMC 5519445. PMID 28108534.
{{cite journal}}: CS1 maint: article number as page number (link) - ^ Yaribash, Shakila; Mohammadi, Keyhan; Sani, Mahmood Alizadeh (2025-03-26). “Alpha-Synuclein Pathophysiology in Neurodegenerative Disorders: A Review Focusing on Molecular Mechanisms and Treatment Advances in Parkinson’s Disease”. Cellular and Molecular Neurobiology. 45 (1). doi:10.1007/s10571-025-01544-2. ISSN 1573-6830. PMC 11947388. PMID 40140103.
- ^ Calabresi, Paolo; Mechelli, Alessandro; Natale, Giuseppina; Volpicelli-Daley, Laura; Di Lazzaro, Giulia; Ghiglieri, Veronica (2023-03-01). “Alpha-synuclein in Parkinson’s disease and other synucleinopathies: from overt neurodegeneration back to early synaptic dysfunction”. Cell Death & Disease. 14 (3): 176. doi:10.1038/s41419-023-05672-9. ISSN 2041-4889.
- ^ Calabresi, Paolo; Mechelli, Alessandro; Natale, Giuseppina; Volpicelli-Daley, Laura; Di Lazzaro, Giulia; Ghiglieri, Veronica (2023-03-01). “Alpha-synuclein in Parkinson’s disease and other synucleinopathies: from overt neurodegeneration back to early synaptic dysfunction”. Cell Death & Disease. 14 (3): 176. doi:10.1038/s41419-023-05672-9. ISSN 2041-4889.
- ^ Burghaus L, Eggers C, Timmermann L, Fink GR, Diederich NJ (February 2012). “Hallucinations in neurodegenerative diseases”. CNS Neurosci Ther (Review). 18 (2): 149–59. doi:10.1111/j.1755-5949.2011.00247.x. PMC 6493408. PMID 21592320.
- ^ Gomperts SN (April 2016). “Lewy body dementias: Dementia with Lewy bodies and Parkinson disease dementia”. Continuum (Minneap Minn) (Review). 22 (2 Dementia): 435–63. doi:10.1212/CON.0000000000000309. PMC 5390937. PMID 27042903.