From Wikipedia, the free encyclopedia
Content deleted Content added
|
|
|||
| Line 65: | Line 65: | ||
|
[[Category:Ethyl esters]] |
[[Category:Ethyl esters]] |
||
|
[[Category:Borate esters]] |
[[Category:Borate esters]] |
||
|
[[Category: |
[[Category:]] |
||
Latest revision as of 19:33, 27 December 2025
Chemical compound
Chemical compound
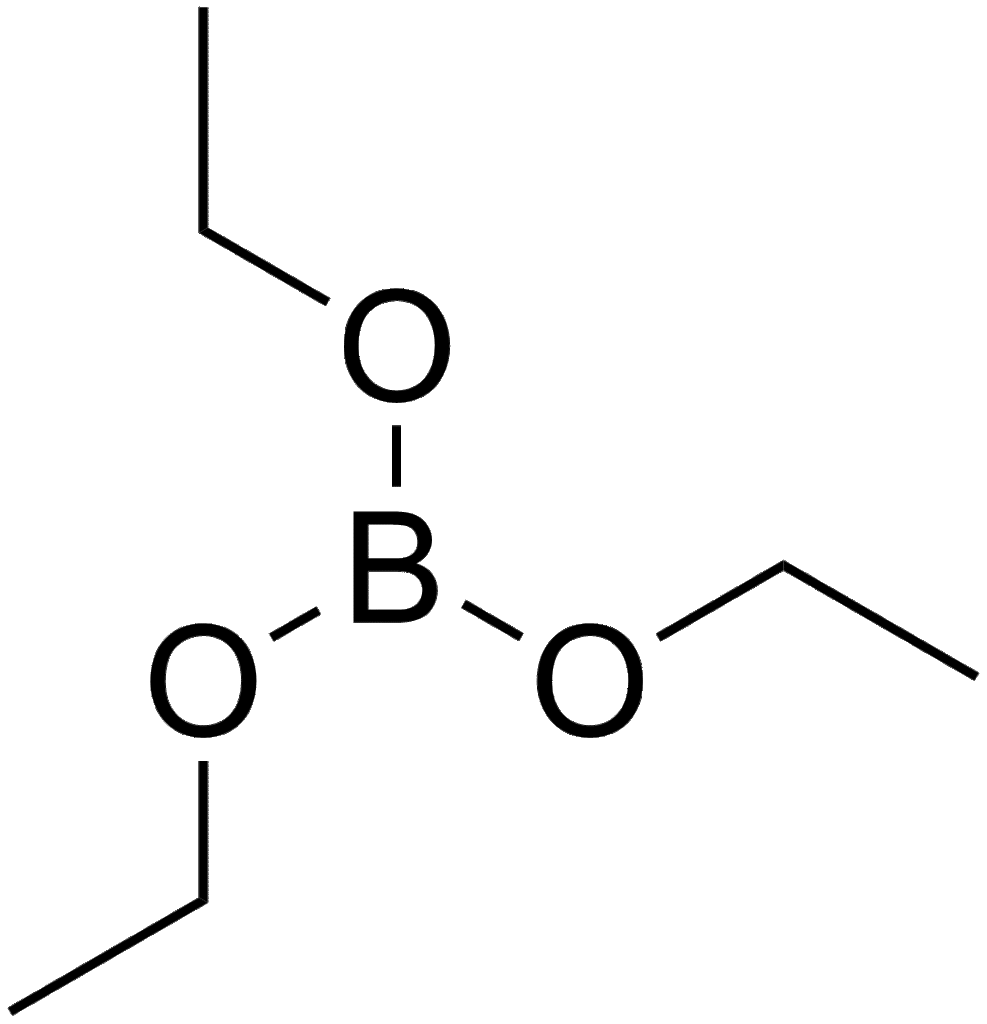
Triethyl borate is a colorless liquid with the formula B(OCH2CH3)3. It is an ester of boric acid and ethanol. It has few applications.[1]
It is a weak Lewis acid (AN = 17 as measured by the Gutmann–Beckett method).[2] It burns with a green flame and solutions of it in ethanol are therefore used in special effects and pyrotechnics.
It is formed by the reaction of boric acid and ethanol in the presence of acid catalyst, where it forms according to the equilibrium reaction:
- B(OH)3 + 3 C2H5OH ⇌ (C2H5O)3B + 3 H2O
In order to increase the rate of forward reaction, the formed water must be removed from reaction media by either azeotropic distillation or adsorption. It is used as a solvent and/or catalyst in preparation of synthetic waxes, resins, paints, and varnishes. It is used as a component of some flame retardants in textile industry and of some welding fluxes.