From Wikipedia, the free encyclopedia
Content deleted Content added
|
|
|||
| Line 2: | Line 2: | ||
|
<!– Images –> |
<!– Images –> |
||
|
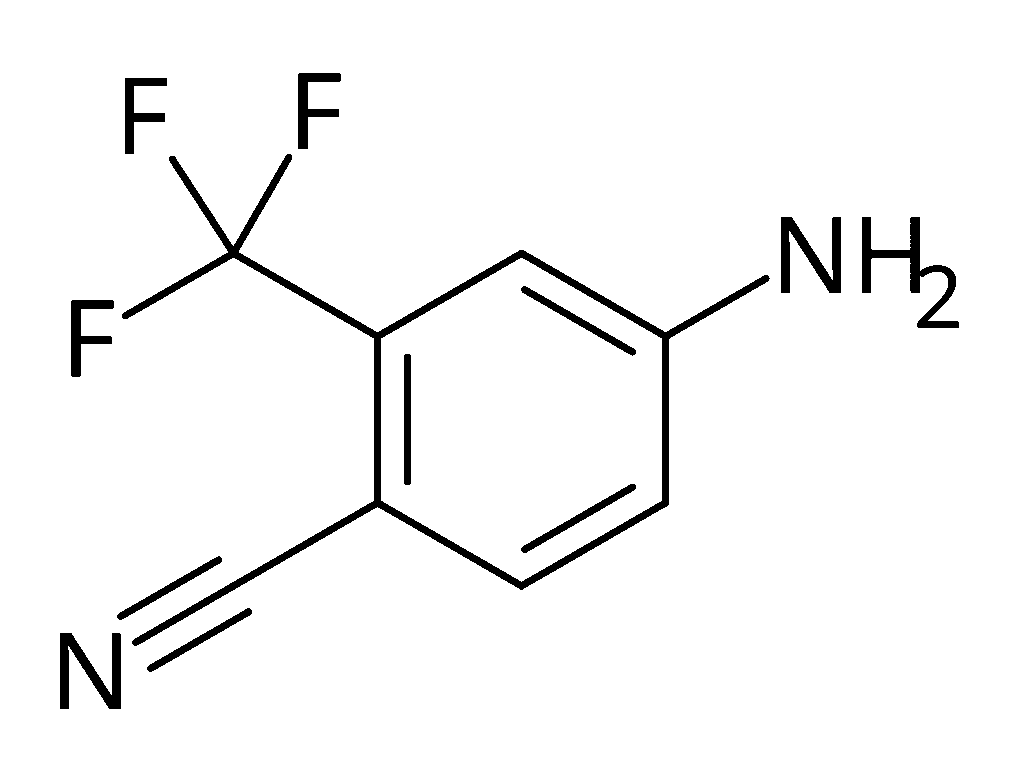
| ImageFile = 4-Cyano-3-(trifluoromethyl)aniline.svg |
| ImageFile = 4-Cyano-3-(trifluoromethyl)aniline.svg |
||
|
| ImageClass = skin-invert-image |
|||
|
| ImageSize = 200px |
| ImageSize = 200px |
||
|
| ImageAlt = |
| ImageAlt = |
||
Latest revision as of 08:27, 23 December 2025
|
|
|
| Names | |
|---|---|
| Preferred IUPAC name
4-Amino-2-(trifluoromethyl)benzonitrile |
|
| Other names
4-Cyano-3-(trifluoromethyl)aniline |
|
| Identifiers | |
| ChemSpider | |
| ECHA InfoCard | 100.109.189 |
| EC Number | |
| UNII | |
|
|
|
|
| Properties | |
| C8H5F3N2 | |
| Molar mass | 186.137 g·mol−1 |
| Hazards | |
| GHS labelling:[1] | |
| Danger | |
| H301, H302, H311, H315, H317, H319, H330, H335 | |
| P260, P261, P262, P264, P264+P265, P270, P271, P272, P280, P284, P301+P316, P301+P317, P302+P352, P304+P340, P305+P351+P338, P316, P319, P320, P321, P330, P332+P317, P333+P317, P337+P317, P361+P364, P362+P364, P403+P233, P405, P501 | |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
|
|
Chemical compound
4-Cyano-3-(trifluoromethyl)aniline, also known as 4-amino-2-(trifluoromethyl)benzonitrile, is a cyanated and trifluoromethylated derivative of aniline. It is the starting material in one of the chemical syntheses of the nonsteroidal antiandrogen bicalutamide.[1]
- ^ Tucker H, Crook JW, Chesterson GJ (1988). “Nonsteroidal antiandrogens. Synthesis and structure-activity relationships of 3-substituted derivatives of 2-hydroxypropionanilides”. Journal of Medicinal Chemistry. 31 (5): 954–9. doi:10.1021/jm00400a011. PMID 3361581.