From Wikipedia, the free encyclopedia
Content deleted Content added
| Line 3: | Line 3: | ||
|
==Selected reactions== |
==Selected reactions== |
||
|
===Benzene oxide=== |
|||
|
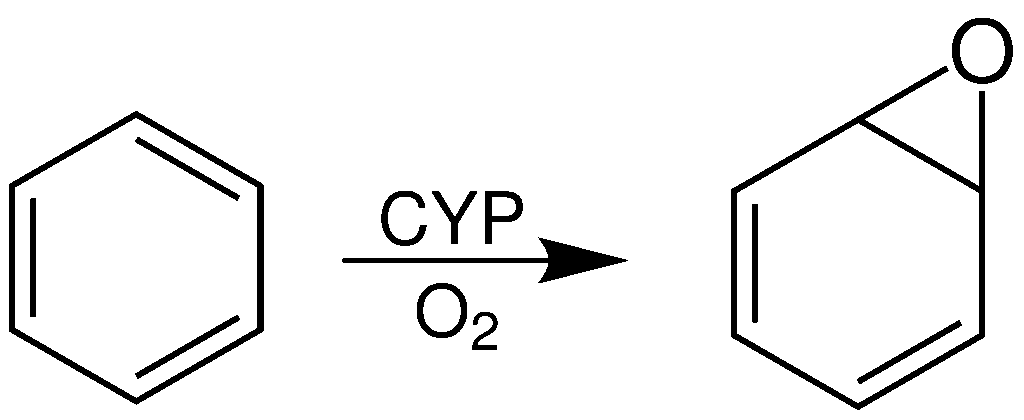
Benzene oxide (C<sub>6</sub>H<sub>6</sub>O) exists as an [[chemical equilibrium|equilibrium]] mixture with the seven-membered [[ring (chemistry)|ring]] [[oxepin]], which has three [[double bond]]s. |
Benzene oxide (C<sub>6</sub>H<sub>6</sub>O) exists as an [[chemical equilibrium|equilibrium]] mixture with the seven-membered [[ring (chemistry)|ring]] [[oxepin]], which has three [[double bond]]s. are [[valence isomer]]s equilibrium via disrotatory 6π ring closing and opening.<ref>{{cite journal | doi = 10.1002/anie.196703851 | journal = Angewandte Chemie International Edition in English | title=Benzene Oxide-Oxepin Valence Tautomerism| volume=6 |issue=5 |pages =385–401 |vauthors = Vogel E, Günther H | year = 1967}}</ref><ref name=”Bansal-1999″>{{cite book |last1=Bansal |first1=Raj K. |title=Heterocyclic Chemistry |date=1999 |publisher=New Age International |location=New Delhi |isbn=9788122412123 |page=378 |edition=3}}</ref> |
||
|
[[File:Oxepin-benzene oxide.png|Oxepin-benzene oxide|center|200px]] |
[[File:Oxepin-benzene oxide.png|Oxepin-benzene oxide|center|200px]] |
||
|
|
[[Benzene oxide]] to give dihydroxydihydrobenzene. The hydration is catalyzed by [[epoxide hydrolase]] enzymes. Dehydration of diols, which is driven by rearomatization, gives phenol. |
||
|
===Naphthalene derivatives=== |
|||
|
Naphthalene-1,2-oxide forms from [[naphthalene]]. It hydrates to give 1,2-dihydroxydihydronaphthalene. The hydration is again catalyzed by [[epoxide hydrolase]] enzymes. Dehydration of this diol gives 1-naphthol. Oxidation of 1,2-dihydroxydihydronaphthalene, catalyzed by dihydrodiol dehydrogenase, gives the [[1,2-naphthoquinone]]. The oxide derived from 1-[[naphthol]] results in 1,4-naphthoquinone. These conversions are relevant to the [[biodegradation]] of naphthalenes.<ref name=”pmid21942631″>{{cite journal | vauthors = Kumagai Y, Shinkai Y, Miura T, Cho AK | title = The chemical biology of naphthoquinones and its environmental implications | journal = Annu. Rev. Pharmacol. Toxicol. | volume = 52 | pages = 221–47 | date = 2012 | pmid = 21942631 | doi = 10.1146/annurev-pharmtox-010611-134517}}</ref> |
|||
|
==References== |
==References== |
||
Revision as of 21:09, 28 January 2026

In chemistry, an arene oxide is an epoxide of an arene. Two important families of arene oxides are benzene oxides and naphthalene oxides as these are intermediates in the oxidative degradation of benzene and naphthalene, two common pollutants.[1] Benzopyrene is also converted to an epoxide, (+)-benzo[a]pyrene-7,8-epoxide.[citation needed]
Selected reactions
Benzene oxide
Benzene oxide (C6H6O) exists as an equilibrium mixture with the seven-membered ring oxepin, which has three double bonds. The arene oxide and oxepin are valence isomers. The equilibrium proceeds via disrotatory 6π ring closing and opening.[2][3]

Benzene oxide hydrates to give dihydroxydihydrobenzene. The hydration is catalyzed by epoxide hydrolase enzymes. Dehydration of this diols, which is driven by rearomatization, gives phenol.
Naphthalene derivatives
Naphthalene-1,2-oxide forms from naphthalene. It hydrates to give 1,2-dihydroxydihydronaphthalene. The hydration is again catalyzed by epoxide hydrolase enzymes. Dehydration of this diol gives 1-naphthol. Oxidation of 1,2-dihydroxydihydronaphthalene, catalyzed by dihydrodiol dehydrogenase, gives the 1,2-naphthoquinone. The oxide derived from 1-naphthol results in 1,4-naphthoquinone. These conversions are relevant to the biodegradation of naphthalenes.[4]
References