The underlying connective tissue attaches to the basal lamina with [[collagen VII]] [[anchoring fibrils]] and [[fibrillin]] [[microfibril]]s.<ref>{{cite journal | vauthors = Paulsson M | title = Basement membrane proteins: structure, assembly, and cellular interactions | journal = Critical Reviews in Biochemistry and Molecular Biology | volume = 27 | issue = 1–2 | pages = 93–127 | year = 1992 | pmid = 1309319 | doi = 10.3109/10409239209082560 | url = http://www.crbmb.com/cgi/content/abstract/27/1/93 | url-status = live | archive-url = https://web.archive.org/web/20071013093529/http://www.crbmb.com/cgi/content/abstract/27/1/93 | archive-date = 2007-10-13 | url-access = subscription }}</ref>
The underlying connective tissue attaches to the basal lamina with [[collagen VII]] [[anchoring fibrils]] and [[fibrillin]] [[microfibril]]s.<ref>{{cite journal | vauthors = Paulsson M | title = Basement membrane proteins: structure, assembly, and cellular interactions | journal = Critical Reviews in Biochemistry and Molecular Biology | volume = 27 | issue = 1–2 | pages = 93–127 | year = 1992 | pmid = 1309319 | doi = 10.3109/10409239209082560 | url = http://www.crbmb.com/cgi/content/abstract/27/1/93 | url-status = live | archive-url = https://web.archive.org/web/20071013093529/http://www.crbmb.com/cgi/content/abstract/27/1/93 | archive-date = 2007-10-13 | url-access = subscription }}</ref>
The basal lamina layer can further be subdivided into two layers based on their visual appearance in electron microscopy. The lighter-colored layer closer to the epithelium is called the [[lamina lucida]], while the denser-colored layer closer to the connective tissue is called the [[lamina densa]]. The [[Electron|electron-dense]] lamina densa layer is about 30–70 [[nanometer]]s thick and consists of an underlying network of reticular [[Type IV collagen|collagen IV]] fibrils which average 30 nanometers in diameter and 0.1–2 [[micrometre|micrometer]]s in thickness and are coated with the heparan sulfate-rich proteoglycan [[perlecan]].<ref>{{cite journal | vauthors = Noonan DM, Fulle A, Valente P, Cai S, Horigan E, Sasaki M, Yamada Y, Hassell JR | display-authors = 6 | title = The complete sequence of perlecan, a basement membrane heparan sulfate proteoglycan, reveals extensive similarity with laminin A chain, low density lipoprotein-receptor, and the neural cell adhesion molecule | journal = The Journal of Biological Chemistry | volume = 266 | issue = 34 | pages = 22939–22947 | date = December 1991 | pmid = 1744087 | doi = 10.1016/S0021-9258(18)54445-8 | doi-access = free }}</ref> In addition to collagen, this supportive matrix contains intrinsic macromolecular components. The lamina lucida layer is made up of [[laminin]], [[integrin]]s, [[entactin]]s, and [[dystroglycan]]s. Integrins are a key component of [[hemidesmosome]]s which serve to anchor the epithelium to the underlying basement membrane.
[[File:Basement membrane organization.jpg|thumb|Organization of basement membrane]]
[[File:Basement membrane organization.jpg|thumb|Organization of basement membrane]]
To represent the above in a visually organised manner, the basement membrane is organized as follows:
* [[epithelium|Epithelial]]/[[mesothelium|mesothelial]]/[[endothelium|endothelial]] tissue (outer layer)
* Basement membrane
** [[Basal lamina]]
*** [[Lamina lucida]]
**** [[laminin]]
**** [[integrin]]s ([[hemidesmosome]]s)
**** [[nidogen]]s
**** [[dystroglycan]]s
*** [[Lamina densa]]
**** [[type IV collagen|collagen IV]] (coated with [[perlecan]], rich in [[heparan sulfate]])
** Attaching proteins (between the basal and reticular laminae)
*** [[Type-VII collagen|collagen VII]] (anchoring fibrils)
*** [[fibrillin]] ([[microfibril]]s)
** [[Reticular fiber|Lamina reticularis]]
*** [[Collagen, type III, alpha 1|collagen III]] (as [[reticular fibers]])
* [[Connective tissue]] ([[Lamina propria]])
== Function ==
== Function ==
Thin fibrous layer between the cells and the adjacent connective tissue in animals
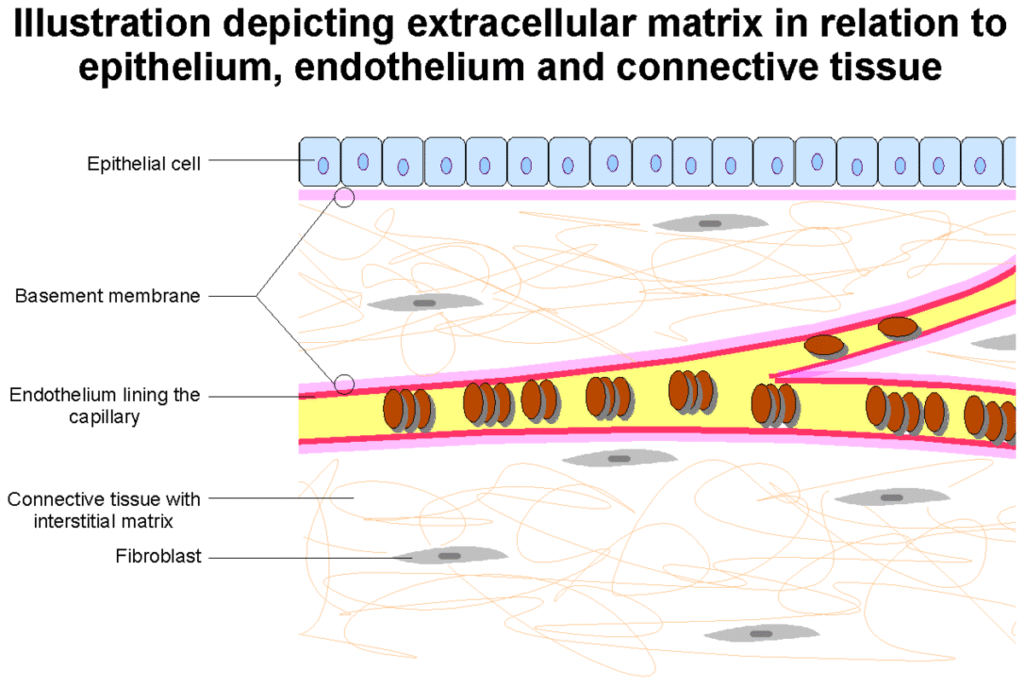
The basement membrane, also known as the basal lamina, is a specialized form of extracellular matrix (ECM) common to all multicellular animals.[1][2] It is a very thin, flexible, and strong sheet-like type of ECM that provides a supporting base for all types of epithelial tissue, separates it from another cell layer such as endothelium, and anchors it to the underlying connective tissue (stroma).[1][3] A basement membrane also surrounds some individual cells, including muscle cells, fat cells, and Schwann cells, separating them from surrounding connective tissue.[1][4] Its composition can vary from tissue to tissue, and even in different regions of the same tissue.[1][4] The other type of ECM is the interstitial matrix.[5]
The basement membrane also acts as a platform for complex cell signaling for polarization, migration, and differentiation.[2][6] It also regulates the exchange of materials between the epithelium and underlying tissues; binds growth factors from the connective tissue to the epithelium that control the development of epithelium.[3] Epithelial cells are pressed closely together having no blood vessels between them but they mostly rest on a bed of loose connective tissue that is rich in blood vessels providing nutrients and removing waste.[3]
Structure
Epithelial cells are polarized. The surface of epithelial cells that face the lumen is the apical surface, and the surface facing the basement membrane is the basal surface.[3] The basement membrane may be described as having two layers or laminae, an external basal lamina, facing the epithelium, and an internal basal lamina that faces the connective tissue.[6] The cells in the internal basal membrane that are closest to the connective tissue show high rates of mitosis, needed to replace skin cell abrasions, and in the GI tract replacement of the cells exposed to digestive enzymes and gastric acid.[3] These two laminae are also known as the basal lamina and the reticular lamina.[7]
The underlying connective tissue attaches to the basal lamina with collagen VII anchoring fibrils and fibrillin microfibrils.[8]

Function



The primary function of the basement membrane is to anchor down the epithelium to its loose connective tissue (stroma) This is achieved by cell-matrix adhesions through cell adhesion molecules.
The basement membrane acts as a mechanical barrier, preventing malignant cells from invading the deeper tissues.[9] Early stages of malignancy that are thus limited to the epithelial layer by the basement membrane are called carcinoma in situ.
The basement membrane is also essential for angiogenesis (development of new blood vessels). Basement membrane proteins have been found to accelerate differentiation of endothelial cells.[10]
The most notable examples of basement membranes are the glomerular basement membrane of the kidney, by the fusion of the basal lamina from the endothelium of glomerular capillaries and the podocyte basal lamina,[11] and the alveolar basement membrane between the alveoli and capillaries of the lung, by the fusion of the basement membrane of the lung alveoli and of the basement membrane of the lung capillaries, which is where oxygen and CO2 diffusion occurs (gas exchange).[4]
As of 2017, other roles for basement membrane include blood filtration and muscle homeostasis.[2] Fractones may be a type of basement membrane, serving as a niche for stem cells.[12][13]
Clinical significance

Some diseases result from a poorly functioning basement membrane. The cause can be genetic defects, injuries by the body’s own immune system, or other mechanisms.[14] Diseases involving basement membranes at multiple locations include:
In histopathology, thickened basement membranes are found in several inflammatory diseases, such as lichen sclerosus, systemic lupus erythematosus or dermatomyositis in the skin, or collagenous colitis in the colon.[17]
Evolutionary origin
These are only found within diploblastic and homoscleromorphic sponge animals. The homoscleromorph were found to be sister to diploblasts in some studies, making the membrane originate once in the history of life. But more recent studies have disregarded diploblast-homoscleromorph group, so other sponges may have lost it (most probable) or the origin in the two groups may be separate.
References
- ^ a b c d Alberts, Bruce (2015). Molecular biology of the cell (Sixth ed.). New York, NY: Garland Science, Taylor and Francis Group. pp. 1068 1069. ISBN 9780815344643.
- ^ a b c Pozzi A, Yurchenco PD, Iozzo RV (January 2017). “The nature and biology of basement membranes”. Matrix Biology. 57–58: 1–11. doi:10.1016/j.matbio.2016.12.009. PMC 5387862. PMID 28040522.
- ^ a b c d e Saladin, Kenneth S. (2011). Human anatomy (3rd ed.). New York: McGraw-Hill. p. 55. ISBN 9780071222075.
- ^ a b c Morrissey, MA; Sherwood, DR (1 May 2015). “An active role for basement membrane assembly and modification in tissue sculpting”. Journal of cell science. 128 (9): 1661–8. doi:10.1242/jcs.168021. PMID 25717004.
- ^ Laurila, Pekka; Leivo, Ilmo (1 January 1993). “Basement membrane and interstitial matrix components form separate matrices in heterokaryons of PYS-2 cells and fibroblasts”. Journal of Cell Science. 104 (1): 59–68. doi:10.1242/jcs.104.1.59.
- ^ a b Sekiguchi R, Yamada KM (2018). “Basement Membranes in Development and Disease”. Current Topics in Developmental Biology. 130: 143–191. doi:10.1016/bs.ctdb.2018.02.005. ISBN 9780128098028. PMC 6701859. PMID 29853176.
- ^ Tortora G, Derrickson B (2012). Principles of anatomy & physiology (13th ed.). Hoboken, NJ: Wiley. pp. 117–118. ISBN 9780470646083.
- ^ Paulsson M (1992). “Basement membrane proteins: structure, assembly, and cellular interactions”. Critical Reviews in Biochemistry and Molecular Biology. 27 (1–2): 93–127. doi:10.3109/10409239209082560. PMID 1309319. Archived from the original on 2007-10-13.
- ^ Liotta LA, Tryggvason K, Garbisa S, Hart I, Foltz CM, Shafie S (March 1980). “Metastatic potential correlates with enzymatic degradation of basement membrane collagen”. Nature. 284 (5751): 67–68. Bibcode:1980Natur.284…67L. doi:10.1038/284067a0. PMID 6243750. S2CID 4356057.
- ^ Kubota Y, Kleinman HK, Martin GR, Lawley TJ (October 1988). “Role of laminin and basement membrane in the morphological differentiation of human endothelial cells into capillary-like structures”. The Journal of Cell Biology. 107 (4): 1589–1598. doi:10.1083/jcb.107.4.1589. PMC 2115245. PMID 3049626.
- ^ “Sect. 7, Ch. 4: Basement Membrane”. Renal Physiology Glomerular Filtration Rate and Renal Blood Flow. Medical College of Georgia, Robert B. Greenblatt, M.D. Library. 1 April 2008. Archived from the original on 1 April 2008. Retrieved 7 May 2018.
{{cite book}}: CS1 maint: bot: original URL status unknown (link) - ^ Mercier F, Kitasako JT, Hatton GI (September 2002). “Anatomy of the brain neurogenic zones revisited: fractones and the fibroblast/macrophage network”. The Journal of Comparative Neurology. 451 (2): 170–188. doi:10.1002/cne.10342. PMID 12209835. S2CID 19919800.
- ^ Sato Y, Kiyozumi D, Futaki S, Nakano I, Shimono C, Kaneko N, et al. (January 2019). Yamashita Y (ed.). “Ventricular-subventricular zone fractones are speckled basement membranes that function as a neural stem cell niche”. Molecular Biology of the Cell. 30 (1): 56–68. doi:10.1091/mbc.E18-05-0286. PMC 6337917. PMID 30379609.
- ^ Henig RM (February 22, 2009). “What’s Wrong With Summer Stiers?”. New York Times. Archived from the original on November 9, 2016.
- ^ Janeway CA (2001). Immunobiology (5th ed.). Garland. ISBN 978-0-8153-3642-6.
- ^ Bardhan A, Bruckner-Tuderman L, Chapple IL, Fine JD, Harper N, Has C, et al. (September 2020). “Epidermolysis bullosa”. Nature Reviews. Disease Primers. 6 (1): 78. doi:10.1038/s41572-020-0210-0. PMID 32973163. S2CID 221861310.
- ^ LeBoit PE (October 2000). “A thickened basement membrane is a clue to…lichen sclerosus!”. The American Journal of Dermatopathology. 22 (5): 457–458. doi:10.1097/00000372-200010000-00014. PMID 11048985.