Inclusion of an additional fused cyclopropane ring at C-16,17 (cf [[cyproterone acetate]]) gives a compound in which progestational activity is said to predominate.
Inclusion of an additional fused cyclopropane ring at C-16,17 (cf [[cyproterone acetate]]) gives a compound in which progestational activity is said to predominate.
[[File:Gestaclone synthesis.svg|thumb|center|700px|Synthesis:<ref>{{cite book | vauthors=((Lednicer, D.)), ((Mitscher, L. A.)) | date= 1980 | title=The organic chemistry of drug synthesis. 2 | publisher=Wiley | isbn=9780471043928}} page 168.</ref> Patent:<ref>GB1095958 idem Friedmund Neumann, Rudolf Wiechert, {{US patent|3441647}} (1969 to Schering Ag).</ref><ref>Gries, Dr Heinz, Hader, Dr Josef, DE1286033 (1969 to Schering Ag).</ref>]]
[[File:Gestaclone synthesis.svg|thumb|center|700px|Synthesis:<ref>{{cite book | vauthors=((Lednicer, D.)), ((Mitscher, L. A.)) | date= 1980 | title=The organic chemistry of drug synthesis. 2 | publisher=Wiley | isbn=9780471043928}} page 168.</ref> Patent:<ref>GB1095958 idem Friedmund Neumann, Rudolf Wiechert, {{US patent|3441647}} (1969 to Schering Ag).</ref><ref>Gries, Dr Heinz, Hader, Dr Josef, DE1286033 (1969 to Schering Ag).</ref>]]
The saponification of the CPA precursor [2701-50-0] (”’1”’) gives the corresponding 17-alcohol [2098-65-9] (”’2”’). Heating in refluxing quinoline results in dehydration with formation of the 16,17-olefin (”’3”’). Reaction with diazomethane gives the pyrazoline (”’4”’), which on heating in acid affords the biscyclopropyl derivative (”’5”’). This compound is then taken on to the 6-chloro analogue by a sequence identical to that used to prepare cyproterone. There is thus obtained the progestin gestaclone (”’6”’).
The saponification of the CPA precursor [2701-50-0] (”’1”’) gives the corresponding 17-alcohol [2098-65-9] (”’2”’). Heating in refluxing quinoline results in dehydration with formation of the 16,17-olefin (”’3”’). Reaction with diazomethane gives the pyrazoline (”’4”’), which on heating in acid affords the biscyclopropyl derivative (”’5”’). This compound is then taken on to the 6-chloro analogue by a sequence identical to that used to prepare cyproterone. There is thus obtained the progestin gestaclone (”’6”’).
==References==
==References==
Chemical compound
Pharmaceutical compound
| ATC code | |
|---|---|
|
|
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Formula | C23H27ClO2 |
| Molar mass | 370.92 g·mol−1 |
| 3D model (JSmol) | |
|
|
|
|
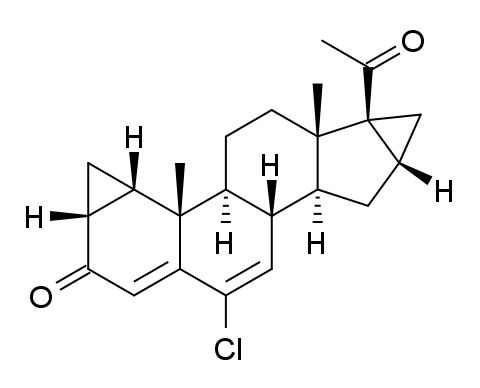
Gestaclone (INN, USAN) (developmental code name SH-1040), also known as 6-chloro-1α,2α:16α,17-bismethylene-4,6-pregnadiene-3,20-dione, is a steroidal progestin of unique chemical structure derived from progesterone that was first described in 1967 and was never marketed.[1]
Inclusion of an additional fused cyclopropane ring at C-16,17 (cf cyproterone acetate) gives a compound in which progestational activity is said to predominate.
The saponification of the CPA precursor [2701-50-0] (1) gives the corresponding 17-alcohol [2098-65-9] (2). Heating in refluxing quinoline results in dehydration with formation of the 16,17-olefin (3). Reaction with diazomethane gives the pyrazoline (4), which on heating in acid affords the biscyclopropyl derivative (5). This compound is then taken on to the 6-chloro analogue by a sequence identical to that used to prepare cyproterone. There is thus obtained the progestin gestaclone (6).
- ^ Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. p. 595. ISBN 978-1-4757-2085-3.
- ^ Lednicer, D., Mitscher, L. A. (1980). The organic chemistry of drug synthesis. 2. Wiley. ISBN 9780471043928. page 168.
- ^ GB1095958 idem Friedmund Neumann, Rudolf Wiechert, U.S. patent 3,441,647 (1969 to Schering Ag).
- ^ Gries, Dr Heinz, Hader, Dr Josef, DE1286033 (1969 to Schering Ag).