From Wikipedia, the free encyclopedia
Content deleted Content added
|
|
|||
| Line 6: | Line 6: | ||
|
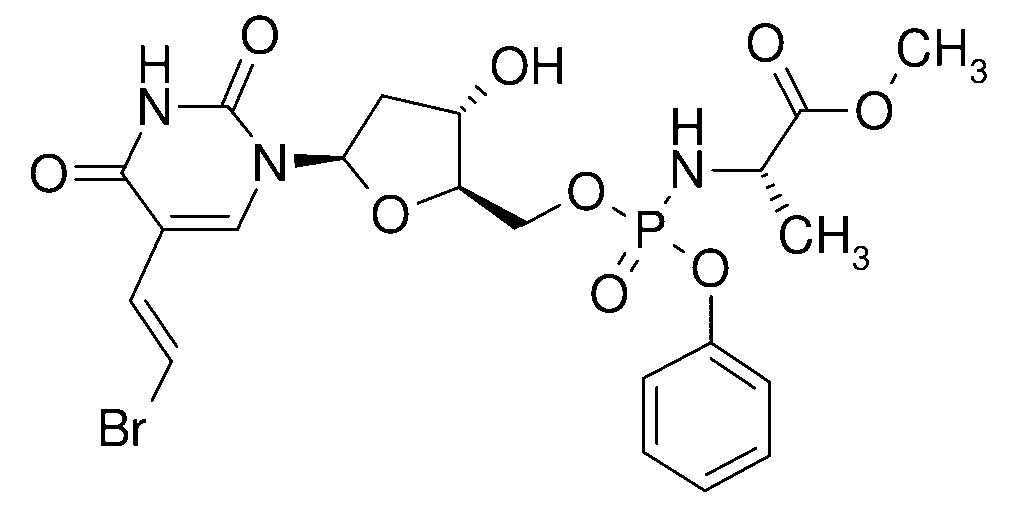
| IUPAC_name = (2”S”)-methyl 2-((((2”R”,3”S”,5”R”)-5-(5-((”E”)-2-bromovinyl)-2,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl)-3-hydroxytetrahydrofuran-2-yl)methoxy)(phenoxy)phosphorylamino)propanoate |
| IUPAC_name = (2”S”)-methyl 2-((((2”R”,3”S”,5”R”)-5-(5-((”E”)-2-bromovinyl)-2,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl)-3-hydroxytetrahydrofuran-2-yl)methoxy)(phenoxy)phosphorylamino)propanoate |
||
|
| image = Thymectacin.svg |
| image = Thymectacin.svg |
||
|
| image_class = skin-invert-image |
|||
|
| alt = Skeletal formula of thymectacin |
| alt = Skeletal formula of thymectacin |
||
|
| width = 260 |
| width = 260 |
||
|
| image2 = Thymectacin-3D-spacefill.png |
| image2 = Thymectacin-3D-spacefill.png |
||
|
| image_class2 = bg-transparent |
|||
|
| alt2 = Space-filling model of the thymectacin molecule |
| alt2 = Space-filling model of the thymectacin molecule |
||
|
| width2 = 240 |
| width2 = 240 |
||
Latest revision as of 17:07, 10 January 2026
Chemical compound
Pharmaceutical compound
 |
|
 |
|
| Other names | NB-1011; NB-101; N-[[5-[(E)-2-Bromovinyl]-2′-deoxyuridin-5′-O-yl]](phenoxy)phosphoryl]-L-alanine methyl ester |
|---|---|
|
|
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Formula | C21H25BrN3O9P |
| Molar mass | 574.321 g·mol−1 |
| 3D model (JSmol) | |
|
|
|
|
| |
|
Thymectacin (NB-1011, NB-101) is an experimental anticancer prodrug of brivudine monophosphate. It is being developed by New Biotics and it entered in phase I clinical trials for colon cancer in 2006.[1]
Thymectacin is a small molecule phosphoramidate derivative of (E)-5-(2-bromovinyl)-2′-deoxyuridine (BVdU) with potential antineoplastic activity.[2] It is selectively active against tumor cells expressing high levels of thymidylate synthase (TS). Thymectacin is converted intracellularly by TS to bromovinyldeoxyuridine monophosphate (BVdUMP) which competes with the natural substrate, deoxyuridine monophosphate, for binding to TS. Unlike TS inhibitors, this agent is a reversible substrate for TS catalysis. Thus, TS retains activity and converts BVdUMP into cytotoxic metabolites.