In general, platinum and palladium complexes are known to efficiently promote catalytic ring opening of silacyclobutanes. Weyenberg and Nelson extensively demonstrated this in 1964 utilizing a platinum metal as a catalyst to [[Hydrosilylation|hydrosilate]] a series of silacyclobutanes (”’1”’) and showed that the reaction involved exchange of Si-H of the hydrosilane and Si-C of silacyclobutane.<ref>{{Cite journal |last=Weyenberg |first=Donald R. |last2=Nelson |first2=Lee E. |date=1965-08-01 |title=Platinum-Catalyzed Reactions of Silacyclobutanes and 1,3-Disilacyclobutanes |url=https://doi.org/10.1021/jo01019a027 |journal=The Journal of Organic Chemistry |volume=30 |issue=8 |pages=2618–2621 |doi=10.1021/jo01019a027 |issn=0022-3263}}</ref> They hypothesized that silacyclobutanes might undergo oxidative-addition with platinum to form 1-platina-2-silacyclobutane. A notable palladium-catalyzed ring opening reaction was reported by Tanaka et. al using acid chloride in the presence of triethylamine in mild contains to produce 1-phenyl-4-(chlorodimethylsilyl)-1-butanone in high yield (”’2”’).<ref name=”:12″>{{Cite journal |last=Tanaka |first=Yoshifumi |last2=Yamashita |first2=Hiroshi |last3=Tanaka |first3=Masato |date=1996-03-19 |title=Palladium- and Platinum-Catalyzed Reactions of Silacyclobutanes with Acid Chlorides Affording Cyclic Silyl Enol Ethers and/or 3-(Chlorosilyl)propyl Ketones |url=https://doi.org/10.1021/om950786n |journal=Organometallics |volume=15 |issue=6 |pages=1524–1526 |doi=10.1021/om950786n |issn=0276-7333}}</ref> Further, these same researchers observed and deciphered the mechanism behind regioselective ring-opening products being obtained in good yield from silacyclobutanes and aryl iodides with a Pd(PPh<sub>3</sub>)<sub>4</sub> catalyst (”’3”’).<ref>{{Cite journal |last=Tanaka |first=Yoshifumi |last2=Nishigaki |first2=Atsushi |last3=Kimura |first3=Yutaka |last4=Yamashita |first4=Masakazu |date=2001 |title=Unexpected regioselectivity in the palladium-catalyzed reaction of silacyclobutanes with aryl iodides |url=https://onlinelibrary.wiley.com/doi/abs/10.1002/aoc.206 |journal=Applied Organometallic Chemistry |language=en |volume=15 |issue=8 |pages=667–670 |doi=10.1002/aoc.206 |issn=1099-0739}}</ref>
In general, platinum and palladium complexes are known to efficiently promote catalytic ring opening of silacyclobutanes. Weyenberg and Nelson extensively demonstrated this in 1964 utilizing a platinum metal as a catalyst to [[Hydrosilylation|hydrosilate]] a series of silacyclobutanes (”’1”’) and showed that the reaction involved exchange of Si-H of the hydrosilane and Si-C of silacyclobutane.<ref>{{Cite journal |last=Weyenberg |first=Donald R. |last2=Nelson |first2=Lee E. |date=1965-08-01 |title=Platinum-Catalyzed Reactions of Silacyclobutanes and 1,3-Disilacyclobutanes |url=https://doi.org/10.1021/jo01019a027 |journal=The Journal of Organic Chemistry |volume=30 |issue=8 |pages=2618–2621 |doi=10.1021/jo01019a027 |issn=0022-3263}}</ref> They hypothesized that silacyclobutanes might undergo oxidative-addition with platinum to form 1-platina-2-silacyclobutane. A notable palladium-catalyzed ring opening reaction was reported by Tanaka et. al using acid chloride in the presence of triethylamine in mild contains to produce 1-phenyl-4-(chlorodimethylsilyl)-1-butanone in high yield (”’2”’).<ref name=”:12″>{{Cite journal |last=Tanaka |first=Yoshifumi |last2=Yamashita |first2=Hiroshi |last3=Tanaka |first3=Masato |date=1996-03-19 |title=Palladium- and Platinum-Catalyzed Reactions of Silacyclobutanes with Acid Chlorides Affording Cyclic Silyl Enol Ethers and/or 3-(Chlorosilyl)propyl Ketones |url=https://doi.org/10.1021/om950786n |journal=Organometallics |volume=15 |issue=6 |pages=1524–1526 |doi=10.1021/om950786n |issn=0276-7333}}</ref> Further, these same researchers observed and deciphered the mechanism behind regioselective ring-opening products being obtained in good yield from silacyclobutanes and aryl iodides with a Pd(PPh<sub>3</sub>)<sub>4</sub> catalyst (”’3”’).<ref>{{Cite journal |last=Tanaka |first=Yoshifumi |last2=Nishigaki |first2=Atsushi |last3=Kimura |first3=Yutaka |last4=Yamashita |first4=Masakazu |date=2001 |title=Unexpected regioselectivity in the palladium-catalyzed reaction of silacyclobutanes with aryl iodides |url=https://onlinelibrary.wiley.com/doi/abs/10.1002/aoc.206 |journal=Applied Organometallic Chemistry |language=en |volume=15 |issue=8 |pages=667–670 |doi=10.1002/aoc.206 |issn=1099-0739}}</ref>
[[File:SilacyclobutanePtPdCatalyst.png|center|thumb|500x500px|Platinum and palladium-catalyzed ring opening reactions with silacyclobutanes.]]In effort to utilize more abundant and affordable catalysts for ring-opening, Oshima et. al demonstrated the viability of nickel-catalyzed ring-opening of silacyclobutanes with aldehydes (”’1”’)<ref name=”:13″>{{Cite journal |last=Hirano |first=Koji |last2=Yorimitsu |first2=Hideki |last3=Oshima |first3=Koichiro |date=2006-02-01 |title=Nickel-Catalyzed Reactions of Silacyclobutanes with Aldehydes: Ring Opening and Ring Expansion Reaction |url=https://doi.org/10.1021/ol0527577 |journal=Organic Letters |volume=8 |issue=3 |pages=483–485 |doi=10.1021/ol0527577 |issn=1523-7060}}</ref> and terminal alkenes (”’2”’),<ref name=”:14″>{{Cite journal |last=Hirano |first=Koji |last2=Yorimitsu |first2=Hideki |last3=Oshima |first3=Koichiro |date=2007-05-01 |title=Nickel-Catalyzed Regio- and Stereoselective Silylation of Terminal Alkenes with Silacyclobutanes: Facile Access to Vinylsilanes from Alkenes |url=https://doi.org/10.1021/ja070938t |journal=Journal of the American Chemical Society |volume=129 |issue=19 |pages=6094–6095 |doi=10.1021/ja070938t |issn=0002-7863}}</ref> retaining stereoselectivity and regioselectivity.
[[File:SilacyclobutanePtPdCatalyst.png|center|thumb|500x500px|Platinum and palladium-catalyzed ring opening reactions with silacyclobutanes.]]In effort to utilize more abundant and affordable catalysts for ring-opening, Oshima et. al demonstrated the viability of nickel-catalyzed ring-opening of silacyclobutanes with aldehydes (”’1”’)<ref name=”:13″>{{Cite journal |last=Hirano |first=Koji |last2=Yorimitsu |first2=Hideki |last3=Oshima |first3=Koichiro |date=2006-02-01 |title=Nickel-Catalyzed Reactions of Silacyclobutanes with Aldehydes: Ring Opening and Ring Expansion Reaction |url=https://doi.org/10.1021/ol0527577 |journal=Organic Letters |volume=8 |issue=3 |pages=483–485 |doi=10.1021/ol0527577 |issn=1523-7060}}</ref> and terminal alkenes (”’2”’),<ref name=”:14″>{{Cite journal |last=Hirano |first=Koji |last2=Yorimitsu |first2=Hideki |last3=Oshima |first3=Koichiro |date=2007-05-01 |title=Nickel-Catalyzed Regio- and Stereoselective Silylation of Terminal Alkenes with Silacyclobutanes: Facile Access to Vinylsilanes from Alkenes |url=https://doi.org/10.1021/ja070938t |journal=Journal of the American Chemical Society |volume=129 |issue=19 |pages=6094–6095 |doi=10.1021/ja070938t |issn=0002-7863}}</ref> retaining stereoselectivity and regioselectivity.
[[File:NiCatalystSilacyclobutane2.png|thumb|Nickel-catalyzed ring-opening of silacyclobutanes with aldehydes (”’1”’)<ref name=”:13″ /> and terminal alkenes (”’2”’).<ref name=”:14″ /> ]]
[[File:NiCatalystSilacyclobutane2.png|thumb|Nickel-catalyzed ring-opening of silacyclobutanes with aldehydes (”’1”’)<ref name=”:13″ /> and terminal alkenes (”’2”’).<ref name=”:14″ /> ]]
=== Transition-Metal-Catalyzed Ring Expansion ===
=== Transition-Metal-Catalyzed Ring Expansion ===
Ring expansion as a means to form a diverse array of sila-heterocycles along with the underlying mechanisms governing this phenomena remain an area of research. In particular, ring expansion using silacyclobutanes has been useful in synthesizing cyclic silly eno.s ethers which are useful precursors in organosilicon chemistry.<ref name=”:0″ /> Ring expansion has been shown to commence with insertion with SO<sub>2</sub>,<ref>{{Cite journal |last=Dubac |first=Jacques |last2=Mazerolles |first2=Pierre |last3=Joly |first3=Monique |date=1978-06-27 |title=Stereochimie de la sulfonation d’organosilanes |url=https://www.sciencedirect.com/science/article/pii/S0022328X00920515 |journal=Journal of Organometallic Chemistry |volume=153 |issue=3 |pages=289–298 |doi=10.1016/S0022-328X(00)92051-5 |issn=0022-328X}}</ref> SO<sub>3</sub>,<ref>{{Cite journal |last=Dubac |first=J. |last2=Mazerolles |first2=P. |last3=Joly |first3=M. |last4=Kitching |first4=W. |last5=Fong |first5=C. W. |last6=Atwell |first6=W. H. |date=1970-11-01 |title=Sultones et sultines organométalliques V. Réactions d’insertion de l’anhydride sulfureux dans la liaison siliciumcarbone: sultines organosiliciques |url=https://www.sciencedirect.com/science/article/pii/S0022328X00861896 |journal=Journal of Organometallic Chemistry |volume=25 |issue=1 |pages=C20–C22 |doi=10.1016/S0022-328X(00)86189-6 |issn=0022-328X}}</ref> phosphorous ylides,<ref>{{Cite journal |last=Schmidbaur |first=Hubert |last2=Wolf |first2=Walter |date=1973 |title=Anormale Silacyclobutan-Ringöffnung durch Alkyliden-trialkylphosphorane |url=https://onlinelibrary.wiley.com/doi/abs/10.1002/ange.19730850807 |journal=Angewandte Chemie |language=en |volume=85 |issue=8 |pages=344–345 |doi=10.1002/ange.19730850807 |issn=1521-3757}}</ref> acid chloride,<ref name=”:12″ /> palladium catalysts,<ref name=”:12″ /> platinum catalysts,<ref name=”:12″ /> nickel catalysts,<ref name=”:13″ /> along with others.<ref name=”:0″ /> Noteable reactions include palladium-catalyzed ring expansion cycloaddition of silacyclobutane with alkynes (”’1”’)<ref name=”:15″>{{Cite journal |last=Chen |first=Ling |last2=Cao |first2=Jian |last3=Xu |first3=Zheng |last4=Zheng |first4=Zhan-Jiang |last5=Cui |first5=Yu-Ming |last6=Xu |first6=Li-Wen |date=2016 |title=Lewis acid catalyzed [2+2] cycloaddition of ynamides and propargyl silyl ethers: synthesis of alkylidenecyclobutenones and their reactivity in ring-opening and ring expansion |url=https://xlink.rsc.org/?DOI=C6CC04648G |journal=Chemical Communications |language=en |volume=52 |issue=61 |pages=9574–9577 |doi=10.1039/C6CC04648G |issn=1359-7345}}</ref> and nickel-catalyzed ring expansion of silacyclobutanes with aldehydes to form cyclic enol ethers (”’2”’)<ref name=”:13″ />.
Ring expansion as a means to form a diverse array of sila-heterocycles along with the underlying mechanisms governing this phenomena remain an area of research. In particular, ring expansion using silacyclobutanes has been useful in synthesizing cyclic silly eno.s ethers which are useful precursors in organosilicon chemistry.<ref name=”:0″ /> Ring expansion has been shown to commence with insertion with SO<sub>2</sub>,<ref>{{Cite journal |last=Dubac |first=Jacques |last2=Mazerolles |first2=Pierre |last3=Joly |first3=Monique |date=1978-06-27 |title=Stereochimie de la sulfonation d’organosilanes |url=https://www.sciencedirect.com/science/article/pii/S0022328X00920515 |journal=Journal of Organometallic Chemistry |volume=153 |issue=3 |pages=289–298 |doi=10.1016/S0022-328X(00)92051-5 |issn=0022-328X}}</ref> SO<sub>3</sub>,<ref>{{Cite journal |last=Dubac |first=J. |last2=Mazerolles |first2=P. |last3=Joly |first3=M. |last4=Kitching |first4=W. |last5=Fong |first5=C. W. |last6=Atwell |first6=W. H. |date=1970-11-01 |title=Sultones et sultines organométalliques V. Réactions d’insertion de l’anhydride sulfureux dans la liaison siliciumcarbone: sultines organosiliciques |url=https://www.sciencedirect.com/science/article/pii/S0022328X00861896 |journal=Journal of Organometallic Chemistry |volume=25 |issue=1 |pages=C20–C22 |doi=10.1016/S0022-328X(00)86189-6 |issn=0022-328X}}</ref> phosphorous ylides,<ref>{{Cite journal |last=Schmidbaur |first=Hubert |last2=Wolf |first2=Walter |date=1973 |title=Anormale Silacyclobutan-Ringöffnung durch Alkyliden-trialkylphosphorane |url=https://onlinelibrary.wiley.com/doi/abs/10.1002/ange.19730850807 |journal=Angewandte Chemie |language=en |volume=85 |issue=8 |pages=344–345 |doi=10.1002/ange.19730850807 |issn=1521-3757}}</ref> acid chloride,<ref name=”:12″ /> palladium catalysts,<ref name=”:12″ /> platinum catalysts,<ref name=”:12″ /> nickel catalysts,<ref name=”:13″ /> along with others.<ref name=”:0″ /> Noteable reactions include palladium-catalyzed ring expansion cycloaddition of silacyclobutane with alkynes (”’1”’)<ref name=”:15″>{{Cite journal |last=Chen |first=Ling |last2=Cao |first2=Jian |last3=Xu |first3=Zheng |last4=Zheng |first4=Zhan-Jiang |last5=Cui |first5=Yu-Ming |last6=Xu |first6=Li-Wen |date=2016 |title=Lewis acid catalyzed [2+2] cycloaddition of ynamides and propargyl silyl ethers: synthesis of alkylidenecyclobutenones and their reactivity in ring-opening and ring expansion |url=https://xlink.rsc.org/?DOI=C6CC04648G |journal=Chemical Communications |language=en |volume=52 |issue=61 |pages=9574–9577 |doi=10.1039/C6CC04648G |issn=1359-7345}}</ref> and nickel-catalyzed ring expansion of silacyclobutanes with aldehydes to form cyclic enol ethers (”’2”’)<ref name=”:13″ />.
[[File:RingOpeningSilacyclobutane.png|thumb|Palladium-catalyzed ring expansion cycloaddition of silacyclobutane with alkynes (”’1”’)<ref name=”:15″ /> and nickel-catalyzed ring expansion of silacyclobutanes with aldehydes to form cyclic enol ethers (”’2”’)<ref name=”:13″ />.]]
[[File:RingOpeningSilacyclobutane.png|thumb|Palladium-catalyzed ring expansion cycloaddition of silacyclobutane with alkynes (”’1”’)<ref name=”:15″ /> and nickel-catalyzed ring expansion of silacyclobutanes with aldehydes to form cyclic enol ethers (”’2”’)<ref name=”:13″ />.]]
Class of chemical compounds
Chemical compound
Silacyclobutane (SCB) or siletane is a four-membered heterocylic ring (silacycle) consisting of one silicon atom and three carbon atoms with the general formula (CH2)3SiH2. Silacyclobutane is one of the most simple molecules in the family of organosilicon compounds. The four-membered ring framework of silacyclobutane is analogous to that of cyclobutane, but one carbon atom is replaced by silicon. All four atoms in silacyclobutane are sp³-hybridized, forming a nonplanar, puckered ring. Derivatives of silacyclobutane are called silacyclobutanes with the general formula (CH2)3SiR2. Since the first synthetic report of silacyclobutane in the 1950’s, silacyclobutanes and its analogues have garnered considerable attention owing to their high ring strain, Lewis acidity, and tunable silicon-carbon bond activation, which enable diverse ring-opening and ring-expansion pathways.[1][2]
Structure
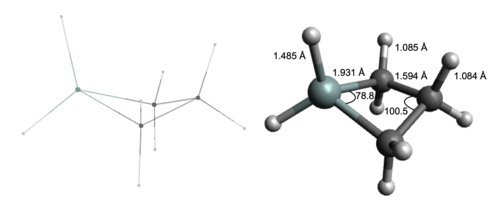
The four carbon atoms in cyclobutane are not coplanar but rather adapts a “puckered” or “butterfly” conformation resulting from the large barrier to internal rotation due to the repulsions of the three adjacent methylene groups.[3] The dihedral angle of the silacyclobutane puckered ring has been calculated as 35.9 ± 2°.[4] For silacyclobutane, the C-Si-C and C-C-C bond angles have been calculated as 78.8° and 100.5°, respectively utilizing multi-configurational self-consistent field (MCSCF).[5]

Like cyclobutane, the small heterocyclic ring of silacyclobutane is highly strained, resulting in lower bond energies when compared to related linear or unstrained, silicon-containing or hydrocarbon rings, such as hexane, cyclohexane, or silacyclohexane. Interestingly, energetic barrier of silacyclobutane[4] (440 cm-1) is lower than that of cyclobutane[6] (498 cm-1), perhaps owing to the greater flexibility of C-Si-C, longer Si–C bonds, and greater bond angle flexibility. Electronic structure calculations of silacyclobutane show that the LUMO is lowered in energy compared to cyclobutane.[7]
The highly strained nature of silacyclobutanes can be investigated through thermolysis, which can provide insights into ring stability and decomposition. Interestingly, there is a considerable difference in liquid- vs. gas-phase thermal decomposition for these compounds.[8] The liquid-phase pyrolysis of 1,1-dimethyl-1-silacyclobutane with the formula (CH2)3SiMe2 results in ring-opening polymerization at 150-200 °C.[9] In comparison, gas-phase pyrolysis of has been demonstrated to primarily undergo unimolecular decomposition to ethane and dimethylsilene at 400-460 °C.[10] It was later shown that there are two minor decomposition pathways; one that forms methyl radicals via the Si–CH3 bond cleavage and another propene–dimethylsilylene species.[11]

Synthesis and History
Fredrick Stanley Kipping, a pioneer of silicon chemistry in the 1920’s was the first to conceptualize silacyclobutane in his series of manuscripts titled “Organic Derivatives of Silicon“[12][13] and is often credited in its history. However, products formed Kipping’s experiments were complex mixtures and he could not conclusively isolate or characterize the silacycles. Kippling’s work was one of the earliest demonstrations that silicon could form stable cyclic structures analogous to carbon rings giving rise to modern organosilicon chemistry.
Grignard Reagent Addition
In 1954, Sommer and Baum reported the first successful synthesis and isolation of a silacyclobutane, 1,1-dimethyl-1-silacyclobutane. Starting from (3-bromopropyl)trimethylsilane, they formed a disiloxane intermediate and subsequently generated a terminal dihalide that was treated with magnesium metal and dilute diethyl ether to form the respective in situ Grignard reagents which perform Wurtz-type coupling intramolecular ring closure by preferentially attacking the bromine site first, followed by the chloride site to form a 4-membered ring.[14]

Classically, silacyclobutanes can be formed using Grignard addition. Notably, this method is employed in the formation of benzosilacyclobutanes from a 1-bromo-2-(bromomethyl)benzene starting material by Gilman and Atwell[15][16] in the 1960’s, followed by de Boer et. al[17] and Kang et. al[18] in the 1980’s.

Halogenated Silacyclobutanes and Nucleophilic Substitution
Vdovin et. al[19] and Laane et. al[20] in the 1960’s synthesized the first halogenated silacyclobutanes, 1,1-dichloro-silacyclobutane and 1,1-difloro-silacyclobutane. The former SiCl2-containing silacyclobutane can be prepared from either (3-bromopropyl)trichlorosilane, which forms more readily, or (3-chloropropyl)trichlorosilane reagents in similar yield. Due to chlorine’s nucleophilic properties, 1,1-dichloro-silacyclobutane proves to be a useful intermediate to accessing further structurally modified silacyclobutanes through nucleophilic substitution.[1] Indeed, as proof of concept, Laane synthesized silacyclobutane-1,1-d2 species using LiAlD4, a deuterated, strong reducing agent analogous to LiAlH4[20] In 1980, Auner and Grobe further expanded the collection of known silacyclobutanes through substitution of the SiCl2-containing silacyclobutane using dimethylamine, Grignard reagents, and sodium cyclopentadienide.[21]

[2+2] Cycloaddition with Alkenes
Alternatively, silacyclobutanes can be prepared through [2+2] cycloaddition reaction with alkenes.[1] Jones et. al demonstrate this using in situ formed vinyldimethylchlorosilane, t-BuLi, and 1,3-butadiene to form a mixture of disilacyclobutanes, monosilacyclobutanes, and silicon-containing products (1).[22][23] When 1,3-butadiene is utilized as a trapping reagent, the reaction is high yielding and E/Z stereochemical products are formed in moderate ratio (2).[24]

Reactivity
Aldol and Allylation Reactions
Strain-Release Lewis Acids
Proposed by Denmark and Sweis in 2002, the term “strain-release Lewis acid” describes the enhanced Lewis acidity of silacyclobutanes that arises from the reduction of ring strain in four-membered ring systems upon coordination by a Lewis base.[2] In silacyclobutane, the ring angle is compressed (79° vs 109° in typical tetrahedron) which poses significant strain on the molecule. Coordination of a fifth ligand lowers the ring strain due to formation of a distorted trigonal bipyramid silicon center (79° vs 90° typical trigonal bipyramidal).[28] This pathway is thermodynamically favored, increasing the tendency of silacyclobutanes to undergo activation by a nucleophile.

Pd-Catalyzed Cross-Coupling
Indeed, the enhanced Lewis acidity of silacyclobutanes has been readily explotited in the cross-coupling reaction of alkenyl silacyclobutanes with alkenyl and aryl halides using an activator, such as TBAF, and palladium catalyst to form the cross-coupling products at room temperature in high yield while retaining olefin geometry of the partners (1).[29] Vinyl and propenyl silacyclobutanes can also be useful cross-coupling partners at moderate conditions (2).[30] Further, aryl silacylobutanes can be used as a cross-coupling reagent to form biaryls, although the reaction typically must be refluxed and tri-tert-butyl added to suppress competing homo-coupling (3).[28]

enoxysila-
cyclobutanes underwent uncatalyzed aldol addition (except
compound 8, Table 1, entry 4) with aldehydes,
Specifically, for
silacyclobutanes, the reaction with nucleophiles allows for relief
of the strain energy via rehybridization of the geometry at silicon
from tetrahedral to trigonal bipyramidal upon formation of a
pentacoordinate species.
Transition-Metal-Catalyzed Ring Opening
The strain-release Lewis acidity of silacyclobutanes can be harnessed in tandem with transition-metal-catalysts to facilitate Si-C bond cleavage and ring-opening. These types of reactions have been leveraged to trigger anionic polymerization in many reported syntheses of functional polymers. Qui-Chao et al. go as far to say silacyclobutanes are, “not only building blocks in organic synthesis but also an emerging class of monomers in polymer chemistry.”[2]
In general, platinum and palladium complexes are known to efficiently promote catalytic ring opening of silacyclobutanes. Weyenberg and Nelson extensively demonstrated this in 1964 utilizing a platinum metal as a catalyst to hydrosilate a series of silacyclobutanes (1) and showed that the reaction involved exchange of Si-H of the hydrosilane and Si-C of silacyclobutane.[31] They hypothesized that silacyclobutanes might undergo oxidative-addition with platinum to form 1-platina-2-silacyclobutane. A notable palladium-catalyzed ring opening reaction was reported by Tanaka et. al using acid chloride in the presence of triethylamine in mild contains to produce 1-phenyl-4-(chlorodimethylsilyl)-1-butanone in high yield (2).[32] Further, these same researchers observed and deciphered the mechanism behind regioselective ring-opening products being obtained in good yield from silacyclobutanes and aryl iodides with a Pd(PPh3)4 catalyst (3).[33]

In effort to utilize more abundant and affordable catalysts for ring-opening, Oshima et. al demonstrated the viability of nickel-catalyzed ring-opening of silacyclobutanes with aldehydes (1)[34] and terminal alkenes (2),[35] retaining stereoselectivity and regioselectivity.

Transition-Metal-Catalyzed Ring Expansion
Ring expansion as a means to form a diverse array of sila-heterocycles along with the underlying mechanisms governing this phenomena remain an area of research. In particular, ring expansion using silacyclobutanes has been useful in synthesizing cyclic silly eno.s ethers which are useful precursors in organosilicon chemistry.[2] Ring expansion has been shown to commence with insertion with SO2,[36] SO3,[37] phosphorous ylides,[38] acid chloride,[32] palladium catalysts,[32] platinum catalysts,[32] nickel catalysts,[34] along with others.[2] Noteable reactions include palladium-catalyzed ring expansion cycloaddition of silacyclobutane with alkynes (1)[39] and nickel-catalyzed ring expansion of silacyclobutanes with aldehydes to form cyclic enol ethers (2)[34].

Photophysical Properties
References
- ^ a b c Huang, Jiapian; Liu, Fei; Wu, Xinyu; Chen, Jian-Qiang; Wu, Jie (2022). “Recent advances in the reactions of silacyclobutanes and their applications”. Organic Chemistry Frontiers. 9 (10): 2840–2855. doi:10.1039/D2QO00410K. ISSN 2052-4129.
- ^ a b c d e Mu, Qiu-Chao; Chen, Jing; Xia, Chun-Gu; Xu, Li-Wen (2018-11-01). “Synthesis of silacyclobutanes and their catalytic transformations enabled by transition-metal complexes”. Coordination Chemistry Reviews. 374: 93–113. doi:10.1016/j.ccr.2018.06.015. ISSN 0010-8545.
- ^ Laane, Jaan (1970-03-01). “Vibrational spectra and normal-coordinate analyses of silacyclobutanes”. Spectrochimica Acta Part A: Molecular Spectroscopy. 26 (3): 517–540. doi:10.1016/0584-8539(70)80099-X. ISSN 0584-8539.
- ^ a b Laane, Jaan; Lord, R. C. (1968-02-15). “Far-Infrared Spectra of Ring Compounds. III. Spectrum, Structure, and Ring-Puckering Potential of Silacyclobutane”. The Journal of Chemical Physics. 48 (4): 1508–1513. doi:10.1063/1.1668871. ISSN 0021-9606.
- ^ a b Gordon, Mark S.; Barton, Thomas J.; Nakano, Haruyuki (1997-12-01). “Ring Opening of Silacyclobutane”. Journal of the American Chemical Society. 119 (49): 11966–11973. doi:10.1021/ja9715354. ISSN 0002-7863.
- ^ Blake, Thomas A.; Xantheas, Sotiris S. (2006-09-01). “Structure, Vibrational Spectrum, and Ring Puckering Barrier of Cyclobutane”. The Journal of Physical Chemistry A. 110 (35): 10487–10494. doi:10.1021/jp062472r. ISSN 1089-5639.
- ^ Krapivin, A.M.; Mägi, M.; Svergun, V.I.; Zaharjan, R.Z.; Babich, E.D.; Ushakov, N.V. (1980-04). “The nmr study and cndo/2 molecular orbital calculation of silacyclobutanes”. Journal of Organometallic Chemistry. 190 (1): 9–33. doi:10.1016/s0022-328x(00)82875-2. ISSN 0022-328X.
- ^ Flowers, M. C.; Gusel’nikov, L. E. (1968). “A kinetic study of the gas-phase thermal decomposition of 1,1-dimethyl-1-silacyclobutane”. Journal of the Chemical Society B: Physical Organic: 419. doi:10.1039/j29680000419. ISSN 0045-6470.
- ^ Nametkin, N. S.; Vdovin, V. M.; Gusel’nikov, L. E.; Zav’yalov, V. I. (1966-03). “Formation of 1,3-disilacyclobutanes in the pyrolytic condensation of 1-silacyclobutanes”. Bulletin of the Academy of Sciences, USSR Division of Chemical Science. 15 (3): 563–563. doi:10.1007/bf00846138. ISSN 0568-5230.
- ^ Gusel’Nikov, L. E.; Flowers, M. C. (1967-01-01). “The thermal decomposition of 1,1-dimethyl-1-silacyclobutane and some reactions of an unstable intermediate containing a silicon–carbon double bond”. Chemical Communications (London) (17): 864–865. doi:10.1039/C19670000864. ISSN 0009-241X.
- ^ Tong, L.; Shi, Y. J. (2011-01). “Gas-phase reaction chemistry of 1,1-dimethyl-1-silacyclobutane as a precursor gas in the hot-wire chemical vapor deposition process — Formation of tetramethylsilane and trimethylsilane”. Canadian Journal of Chemistry. 89 (1): 19–26. doi:10.1139/V10-147. ISSN 0008-4042.
- ^ Kipping, Frederic Stanley (1912-01-01). “CCXXIV.—Organic derivatives of silicon. Part XVII. Some condensation products of diphenylsilicanediol”. Journal of the Chemical Society, Transactions. 101 (0): 2125–2142. doi:10.1039/CT9120102125. ISSN 0368-1645.
- ^ Kipping, Frederic Stanley; Sands, James Edwin (1921-01-01). “XCIV.—Organic derivatives of silicon. Part XXVI. Piperidine as an analytical reagent”. Journal of the Chemical Society, Transactions. 119 (0): 848–850. doi:10.1039/CT9211900848. ISSN 0368-1645.
- ^ Sommer, Leo H.; Baum, Gerald A. (1954-10). “A SILICON-CONTAINING 4-RING”. Journal of the American Chemical Society. 76 (19): 5002–5002. doi:10.1021/ja01648a083. ISSN 0002-7863.
- ^ Gilman, Henry.; Atwell, William H. (1964-07-01). “Small-Ring Organosilicon Compounds. I. A Comparison of the Reactivities of 1,1,2-Triphenyl-1-silacyclobutane and 1,1,2-Triphenyl-1-silacyclopentane”. Journal of the American Chemical Society. 86 (13): 2687–2693. doi:10.1021/ja01067a035. ISSN 0002-7863.
- ^ Gilman, Henry.; Atwell, William H. (1964-07-01). “Small-Ring Organosilicon Compounds. I. A Comparison of the Reactivities of 1,1,2-Triphenyl-1-silacyclobutane and 1,1,2-Triphenyl-1-silacyclopentane”. Journal of the American Chemical Society. 86 (13): 2687–2693. doi:10.1021/ja01067a035. ISSN 0002-7863.
- ^ de Boer, H. J. R.; Akkerman, O. S.; Bickelhaupt, F. (1987-04-10). “An investigation of the di-Grignard approach to metallabenzocyclobutenes of group 14”. Journal of Organometallic Chemistry. 321 (3): 291–306. doi:10.1016/0022-328X(87)80301-7. ISSN 0022-328X.
- ^ Kang, Kyung-Tae; Song, Hwan-Young; Seo, Hee-Chan (1985-05-05). “SYNTHESIS OF 4-SUBSTITUTED 1,1-DI- t -BUTYLBENZOSILACYCLOBUTENES”. Chemistry Letters. 14 (5): 617–620. doi:10.1246/cl.1985.617. ISSN 0366-7022.
- ^ Nametkin, N. S.; Vdovin, V. M.; Grinberg, P. L. (1964-06). “Telomerization of silicacyclobutanes”. Bulletin of the Academy of Sciences, USSR Division of Chemical Science. 13 (6): 1055–1055. doi:10.1007/bf01141674. ISSN 0568-5230.
- ^ a b c Laane, J. (1967-03). “Synthesis of Silacyclobutane and Some Related Compounds”. Journal of the American Chemical Society. 89 (5): 1144–1147. doi:10.1021/ja00981a020. ISSN 0002-7863.
- ^ a b Auner, N.; Grobe, J. (1980-04-01). “Silaethene: I. Darstellung und charakterisierung von monosilacyclobutanen”. Journal of Organometallic Chemistry. 188 (1): 25–52. doi:10.1016/S0022-328X(00)83695-5. ISSN 0022-328X.
- ^ a b Jones, Paul Ronald; Lim, Thomas F. O. (1977-12-01). “Silaethylene intermediates from .alpha.-lithiosilanes. 2. Reactions with chlorosilanes and 1,3-butadiene”. Journal of the American Chemical Society. 99 (26): 8447–8451. doi:10.1021/ja00468a011. ISSN 0002-7863.
- ^ a b Jones, Paul Ronald; Lim, Thomas F. O.; Pierce, Richard A. (1980-07-01). “.alpha.-Lithiosilanes. 4. Silaethylene cycloadditions with conjugated dienes”. Journal of the American Chemical Society. 102 (15): 4970–4973. doi:10.1021/ja00535a025. ISSN 0002-7863.
- ^ a b Sewald, Norbert; Ziche, Wolfgang; Wolff, Astrid; Auner, Norbert (1993-10-01). “Silaheterocycles. 26. Facile synthesis of silicon dichloro substituted 3-vinyl-1-silacyclobutanes from silene/butadiene [2+2] cycloaddition reactions: model compounds for vinylsilacyclobutane .fwdarw. silacyclohexene rearrangements”. Organometallics. 12 (10): 4123–4134. doi:10.1021/om00034a055. ISSN 0276-7333.
- ^ Myers, Andrew G.; Kephart, Susan E.; Chen, Hou (1992-09-01). “Silicon-directed aldol reactions. Rate acceleration by small rings”. Journal of the American Chemical Society. 114 (20): 7922–7923. doi:10.1021/ja00046a054. ISSN 0002-7863.
- ^ Denmark, Scott E.; Griedel, Brian D.; Coe, Diane M.; Schnute, Mark E. (1994-08-01). “Chemistry of Enoxysilacyclobutanes: Highly Selective Uncatalyzed Aldol Additions”. Journal of the American Chemical Society. 116 (16): 7026–7043. doi:10.1021/ja00095a004. ISSN 0002-7863.
- ^ Matsumoto, Kozo; Oshima, Koichiro; Utimoto, Kiitiro (1994-11-01). “Noncatalyzed Stereoselective Allylation of Carbonyl Compounds with Allylsilacyclobutanes”. The Journal of Organic Chemistry. 59 (23): 7152–7155. doi:10.1021/jo00102a052. ISSN 0022-3263.
- ^ a b c d Denmark, Scott E.; Sweis, Ramzi F. (2002-06-18). “Design and Implementation of New, Silicon-Based, Cross-Coupling Reactions: Importance of Silicon−Oxygen Bonds”. Accounts of Chemical Research. 35 (10): 835–846. doi:10.1021/ar020001r. ISSN 0001-4842.
- ^ a b Denmark, Scott E.; Choi, Jun Young (1999-06-01). “Highly Stereospecific, Cross-Coupling Reactions of Alkenylsilacyclobutanes”. Journal of the American Chemical Society. 121 (24): 5821–5822. doi:10.1021/ja9908117. ISSN 0002-7863.
- ^ a b Denmark, Scott E.; Wang, Zhigang (2000). “1-Methyl-1-vinyl- and 1-Methyl-1-(prop-2-enyl)silacyclobutane: Reagents for Palladium-Catalyzed Cross-Coupling Reactions of Aryl Halides”. Synthesis. 2000 (07): 999–1003. doi:10.1055/s-2000-6292. ISSN 0039-7881.
- ^ Weyenberg, Donald R.; Nelson, Lee E. (1965-08-01). “Platinum-Catalyzed Reactions of Silacyclobutanes and 1,3-Disilacyclobutanes”. The Journal of Organic Chemistry. 30 (8): 2618–2621. doi:10.1021/jo01019a027. ISSN 0022-3263.
- ^ a b c d Tanaka, Yoshifumi; Yamashita, Hiroshi; Tanaka, Masato (1996-03-19). “Palladium- and Platinum-Catalyzed Reactions of Silacyclobutanes with Acid Chlorides Affording Cyclic Silyl Enol Ethers and/or 3-(Chlorosilyl)propyl Ketones”. Organometallics. 15 (6): 1524–1526. doi:10.1021/om950786n. ISSN 0276-7333.
- ^ Tanaka, Yoshifumi; Nishigaki, Atsushi; Kimura, Yutaka; Yamashita, Masakazu (2001). “Unexpected regioselectivity in the palladium-catalyzed reaction of silacyclobutanes with aryl iodides”. Applied Organometallic Chemistry. 15 (8): 667–670. doi:10.1002/aoc.206. ISSN 1099-0739.
- ^ a b c d e Hirano, Koji; Yorimitsu, Hideki; Oshima, Koichiro (2006-02-01). “Nickel-Catalyzed Reactions of Silacyclobutanes with Aldehydes: Ring Opening and Ring Expansion Reaction”. Organic Letters. 8 (3): 483–485. doi:10.1021/ol0527577. ISSN 1523-7060.
- ^ a b Hirano, Koji; Yorimitsu, Hideki; Oshima, Koichiro (2007-05-01). “Nickel-Catalyzed Regio- and Stereoselective Silylation of Terminal Alkenes with Silacyclobutanes: Facile Access to Vinylsilanes from Alkenes”. Journal of the American Chemical Society. 129 (19): 6094–6095. doi:10.1021/ja070938t. ISSN 0002-7863.
- ^ Dubac, Jacques; Mazerolles, Pierre; Joly, Monique (1978-06-27). “Stereochimie de la sulfonation d’organosilanes”. Journal of Organometallic Chemistry. 153 (3): 289–298. doi:10.1016/S0022-328X(00)92051-5. ISSN 0022-328X.
- ^ Dubac, J.; Mazerolles, P.; Joly, M.; Kitching, W.; Fong, C. W.; Atwell, W. H. (1970-11-01). “Sultones et sultines organométalliques V. Réactions d’insertion de l’anhydride sulfureux dans la liaison siliciumcarbone: sultines organosiliciques”. Journal of Organometallic Chemistry. 25 (1): C20 – C22. doi:10.1016/S0022-328X(00)86189-6. ISSN 0022-328X.
- ^ Schmidbaur, Hubert; Wolf, Walter (1973). “Anormale Silacyclobutan-Ringöffnung durch Alkyliden-trialkylphosphorane”. Angewandte Chemie. 85 (8): 344–345. doi:10.1002/ange.19730850807. ISSN 1521-3757.
- ^ a b Chen, Ling; Cao, Jian; Xu, Zheng; Zheng, Zhan-Jiang; Cui, Yu-Ming; Xu, Li-Wen (2016). “Lewis acid catalyzed [2+2] cycloaddition of ynamides and propargyl silyl ethers: synthesis of alkylidenecyclobutenones and their reactivity in ring-opening and ring expansion”. Chemical Communications. 52 (61): 9574–9577. doi:10.1039/C6CC04648G. ISSN 1359-7345.